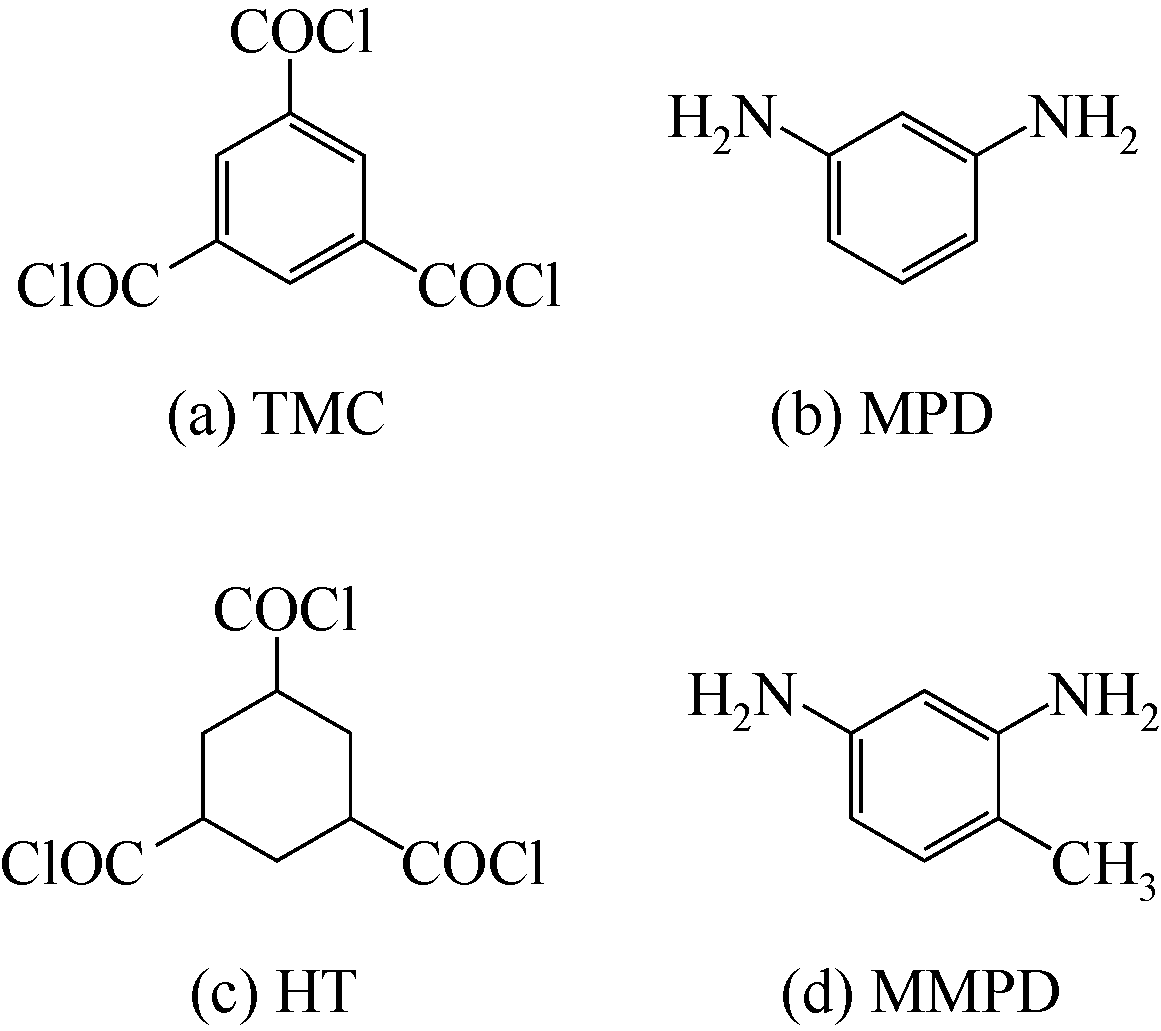
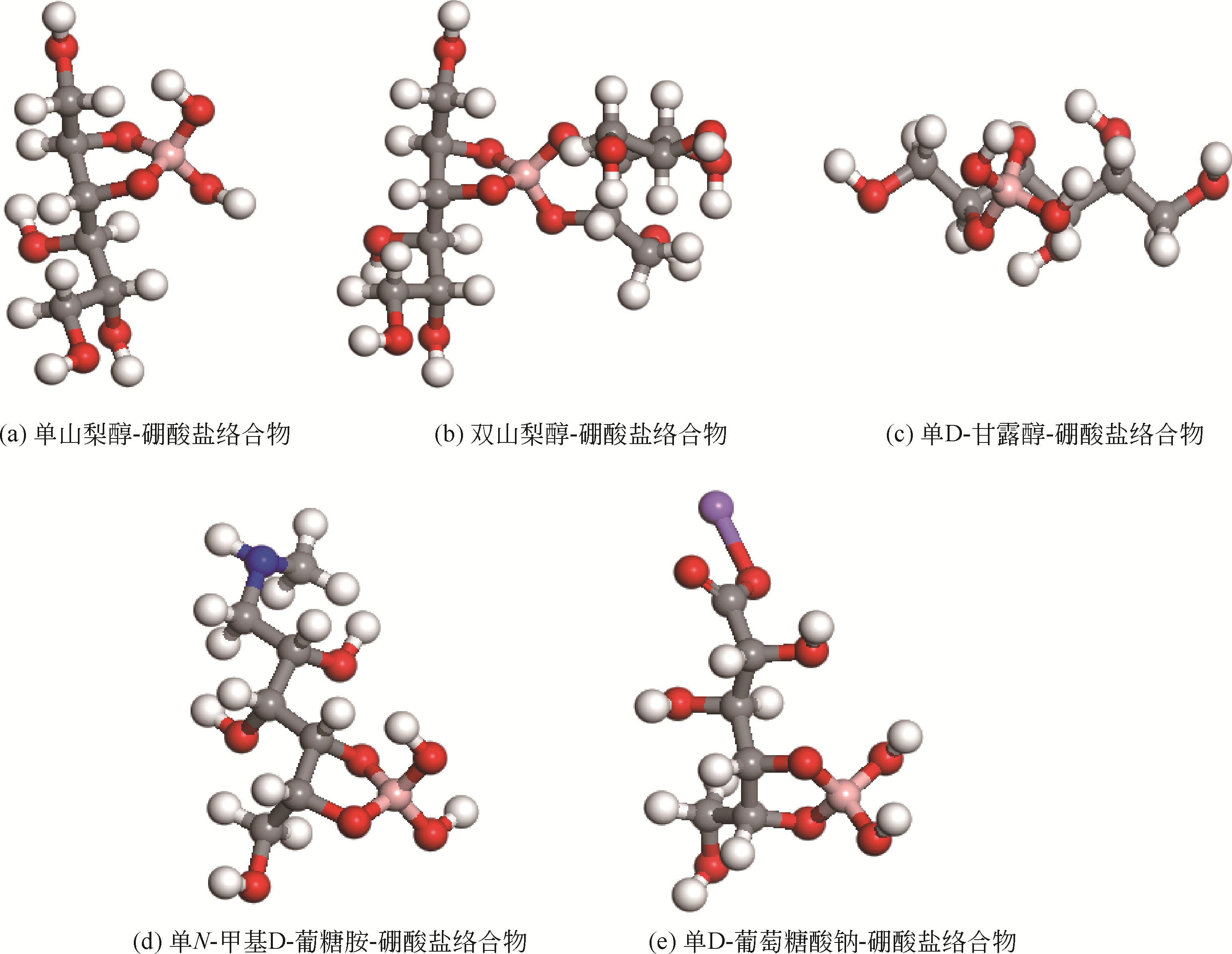
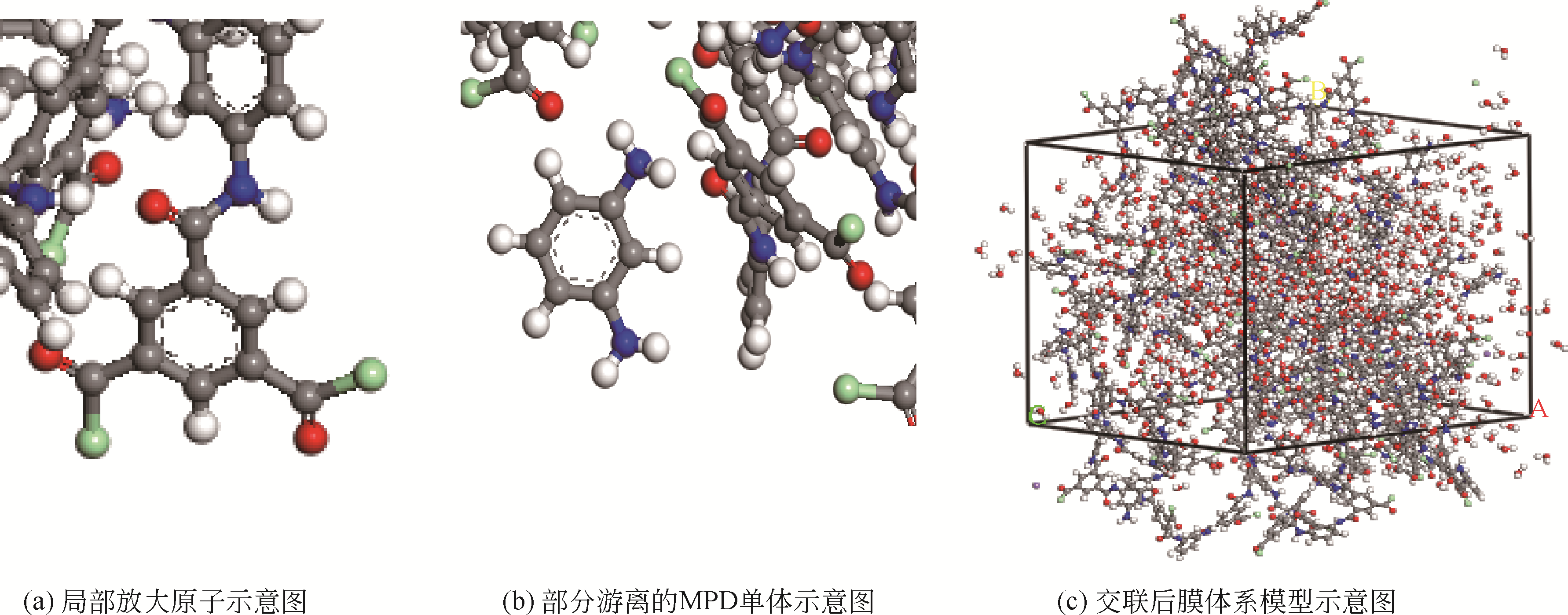
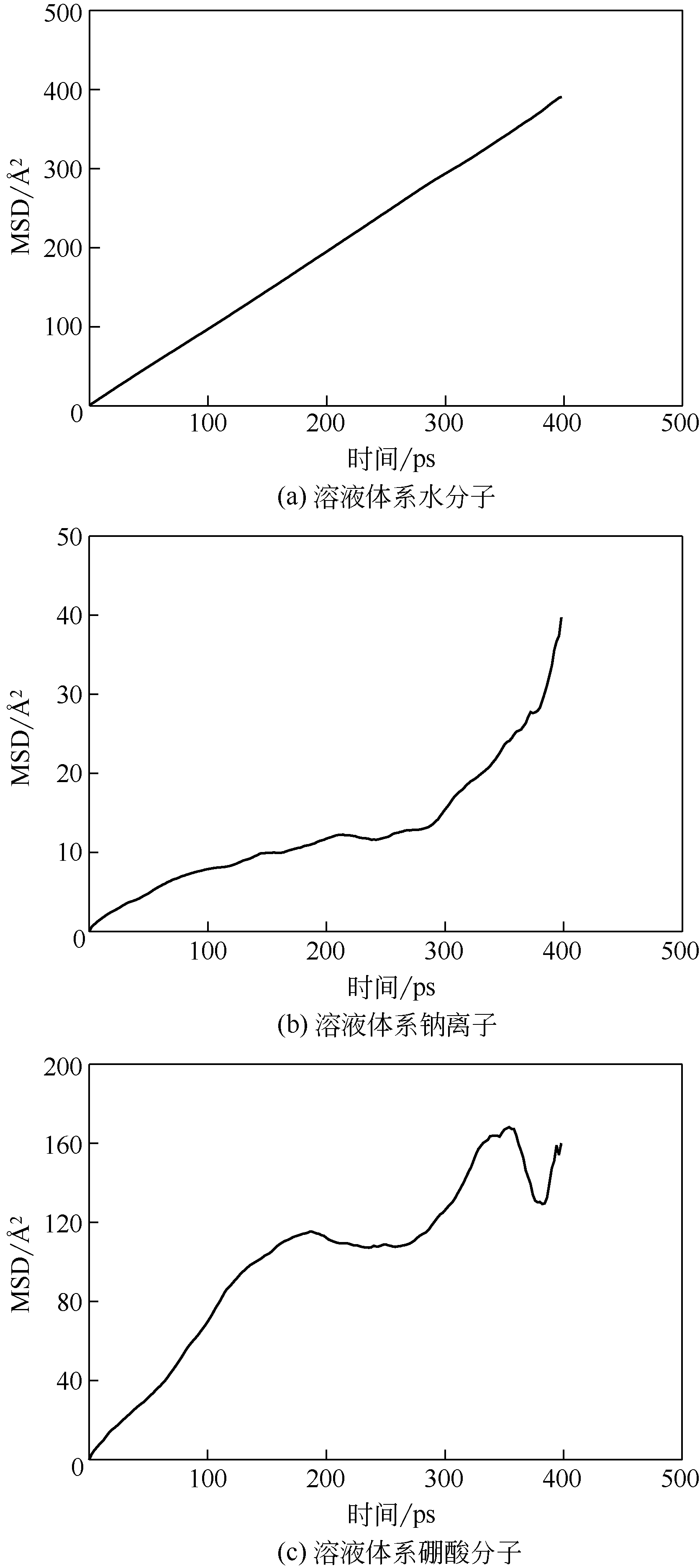
| 1 |
DING M X , GHOUFI A , SZYMCZYK A . Molecular simulations of polyamide reverse osmosis membranes[J]. Desalination, 2014,343(12): 48-53.
|
| 2 |
JIN X , TANG C Y , GU Y S , et al . Boric acid permeation in forward osmosis membrane processes: modeling, experiments, and implications[J]. Environmental Science & Technology, 2011, 45: 2323–2330.
|
| 3 |
GULER E , KAYA C , KABAY N , et al . Boron removal from seawater: state-of-the-art review[J]. Desalination, 2015, 356: 85–93.
|
| 4 |
OPHEK L , BIMHACK L , NIR O, et al . Reducing the specific energy consumption of 1st-pass SWRO by application of high-flux membranes fed with high-pH,decarbonated seawater[J]. Water Research, 2015, 85: 185-192.
|
| 5 |
DYDO P . Transport model for boric acid, monoborate and borate complexes across thin-film composite reverse osmosis membrane[J]. Desalination, 2013, 311: 69–79.
|
| 6 |
隋红,李琳, 陈道毅,等 .分子模拟多溴联苯醚在石英砂纳米孔中的吸附行为[J]. 化工进展,2015,34(9): 3444-3451.
|
|
SUI H, LI L , CHEN D Y , et al . Molecular dynamic simulation of adsorption of polybrominated diphenyl ethers in silica nanopores[J]. Chemical Industry and Engineering Progress, 2015, 34(9): 3444-3451.
|
| 7 |
SHEN M , KETEN S , LUEPTOW R M . Dynamics of water and solute transport in polymeric reverse osmosis membranes via molecular dynamics simulations[J]. J. Membr. Sci., 2016, 506: 95-108.
|
| 8 |
HUGHES Z E , GALE J D . A computational investigation of the properties of a reverse osmosis membrane[J]. J. Mater. Chem., 2010, 20(36): 7788-7799.
|
| 9 |
WEI T , ZHANG L , ZHAO H Y , et al . Aromatic polyamide reverse osmosis membrane:an atomistic molecular dynamic simulation[J]. J. Phys. Chem. B, 2016, 120(39): 10311-10318.
|
| 10 |
GAO W M , SHE F H , ZHANG J , et al . Understanding water and ion transport behaviour and permeability through poly(amide) thin film composite membrane[J]. J. Membr. Sci., 2015, 487: 32-39.
|
| 11 |
LUO Y , HARDER E , FAIBISH R S , et al . Computer simulations of water flux and salt permeability of the reverse osmosis FT-30 aromatic polyamide membrane[J]. J. Membr. Sci., 2011, 384(1): 1-9.
|
| 12 |
刘清芝 . 反渗透膜及水溶液内扩散过程的分子模拟研究[D]. 青岛: 中国海洋大学, 2007.
|
|
LIU Q Z . Molecular simulation study of diffusion process in reverse osmosis membrane and aqueous solution[D]. Qingdao: Ocean University of China, 2007.
|
| 13 |
刘清芝, 杨登峰, 胡仰栋 . 水和盐分子在反渗透膜内扩散过程的分子模拟[J]. 高等学校化学学报, 2009, 30(3): 568-572.
|
|
LIU Q Z , YANG D F , HU Y D . Molecular simulation of diffusion of water and salt molecules in reverse osmosis membrane[J]. Chemical Journal of Chinese Universities, 2009, 30(3): 568-572.
|
| 14 |
GEFFEN N , SEMIAT R , EISEN M S , et al . Boron removal from water by complexation to polyol compounds[J]. J. Membr. Sci., 2006, 286: 45-51.
|
| 15 |
TU K L , CHIVAS A R , NGHIEM L D . Enhanced boron rejection by NF/RO membranes by complexation with polyols: measurement and mechanisms[J]. Desalination, 2013, 310: 115-121.
|
| 16 |
DING M X , SZYMCZYK A , GOUJON F , et al . Structure and dynamics of water confined in a polyamide reverse-osmosis membrane:a molecular-simulation study[J]. J. Membr. Sci., 2014, 458: 236-244.
|
| 17 |
赵海洋, 张林 . 聚酰胺反渗透膜的分子动力学模拟研究进展[J]. 化工进展, 2017, 36(12): 4319-4328.
|
|
ZHAO H Y , ZHANG L . Research progress in molecular dynamics simulation of polyamide reverse osmosis membrane[J]. Chemical Industry and Engineering Progress, 2017, 36(12): 4319-4328.
|
| 18 |
朱绘丽 . 水和盐分子在反渗透膜中溶解扩散过程的分子模拟[D]. 青岛: 中国海洋大学, 2011.
|
|
ZHU H L . Molecular simulation of dissolution and diffusion of water and salt molecules in reverse osmosis membrane[D]. Qingdao: Ocean University of China, 2011.
|
| 19 |
KOLEV V , FREGER V . Hydration, porosity and water dynamics in the polyamide layer of reverse osmosis membranes: a molecular dynamics study[J]. Polymer, 2014, 55: 1420-1426.
|
| 20 |
LIU Q Z , HU Y D . Application of the clustering method in molecular dynamics simulation of the diffusion coefficient[J]. J. Ocean Univ. Chin., 2008, 7(1): 43-47.
|
| 21 |
ABASCAL J , VEGA C . A general purpose model for the condensed phases of water:TIP4P/2005[J]. J. Chem. Phys., 2005, 123(23): 234505.
|
| 22 |
KOTELYANSKII M J , WAGNER N J , PAULAITIS M E . Molecular dynamics simulation study of the mechanisms of water diffusion in a hydrated, amorphous polyamide[J]. Computational and Theoretical Polymer Science, 1999, 9: 301-306.
|
 ),Weixue ZHANG,Yawei DU(
),Weixue ZHANG,Yawei DU( )
)