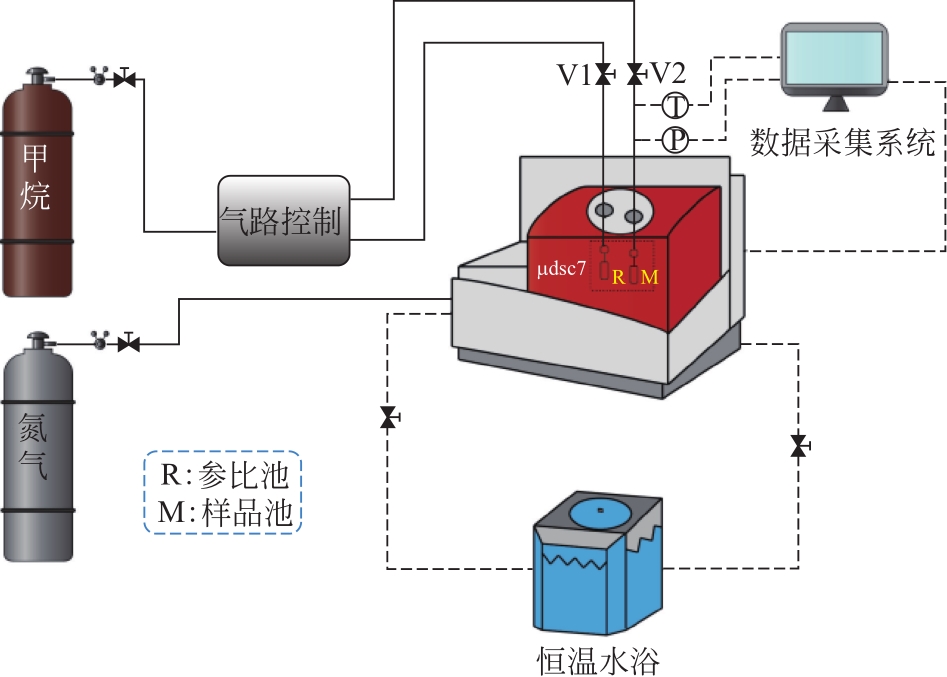
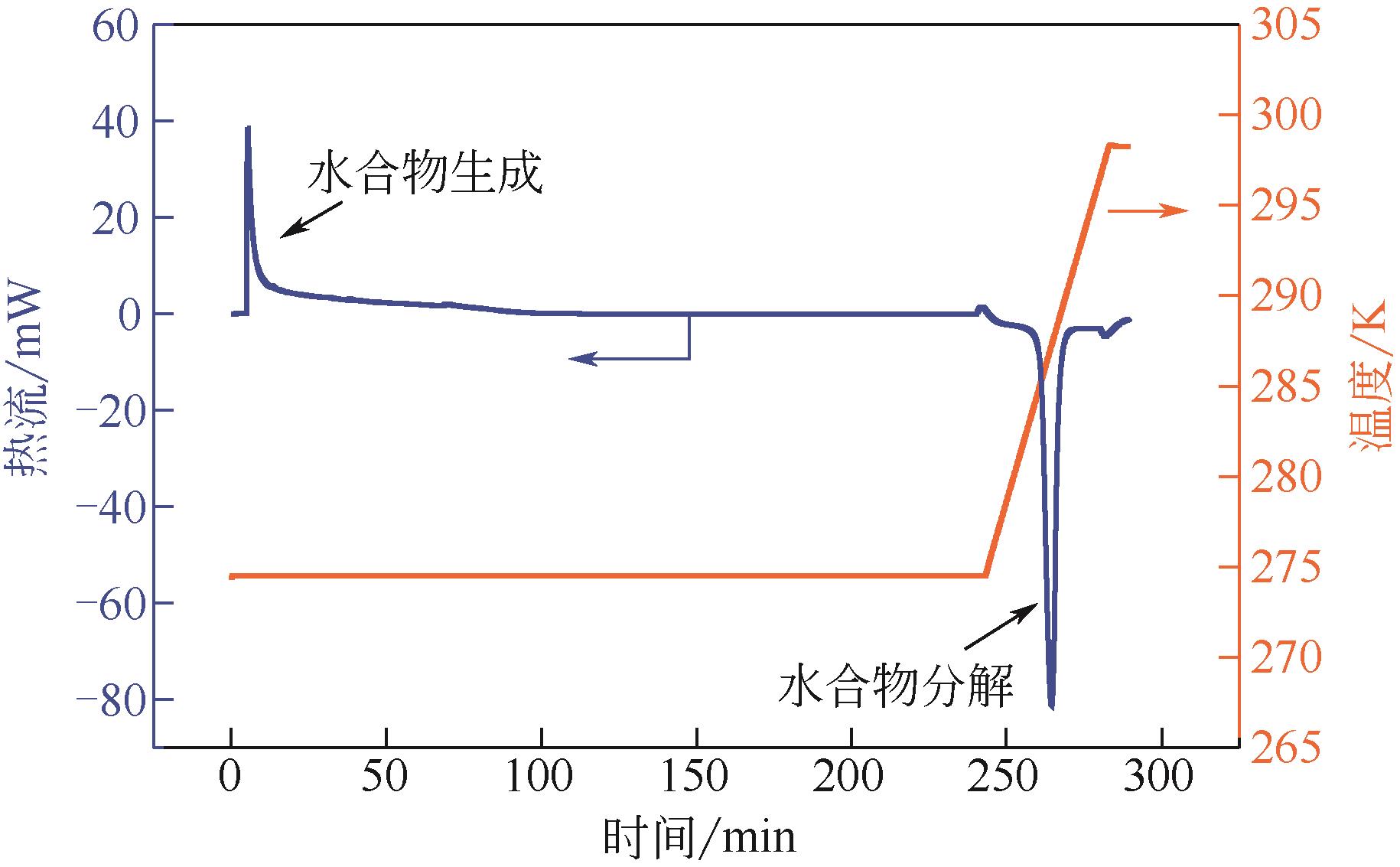
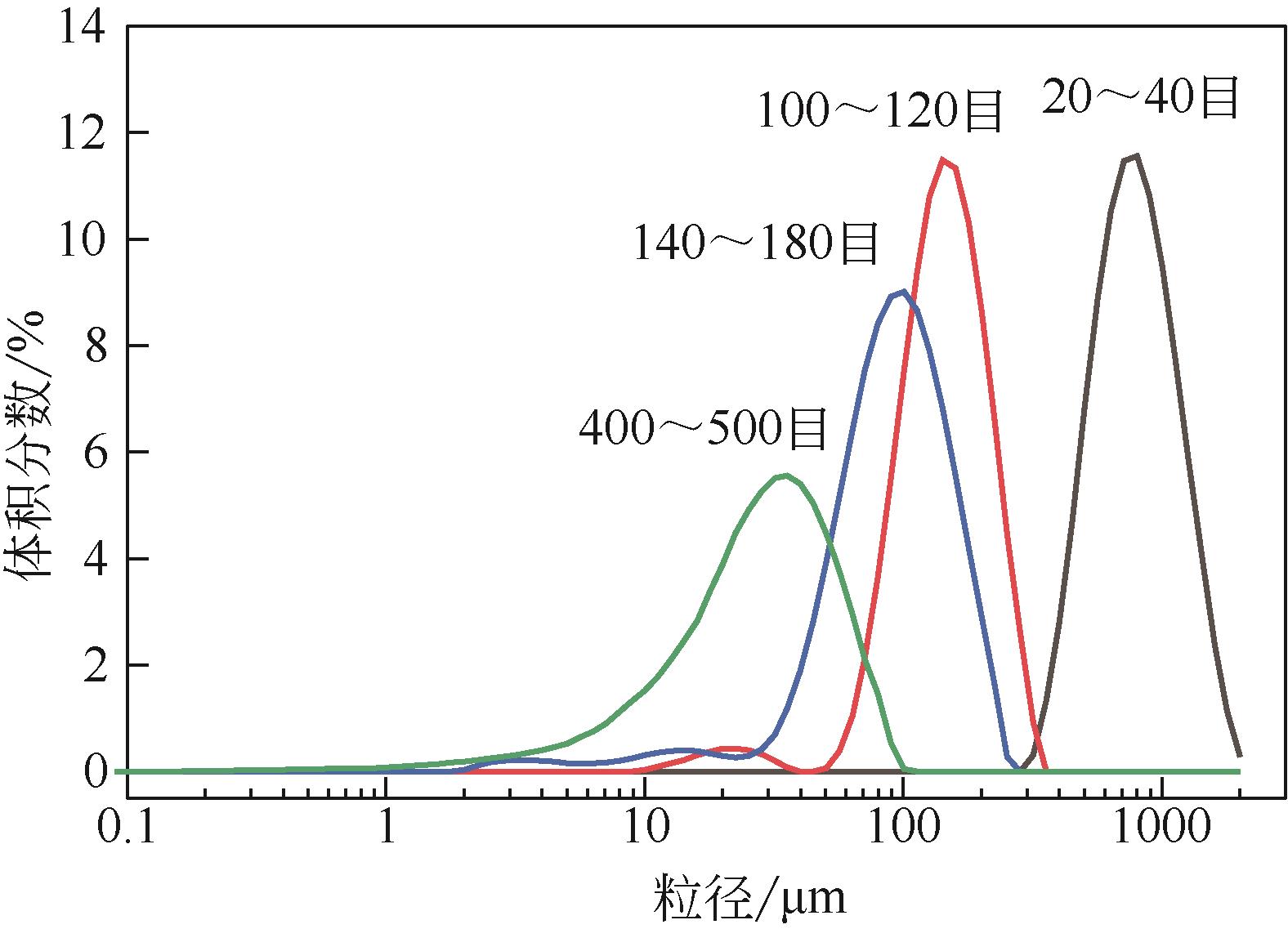
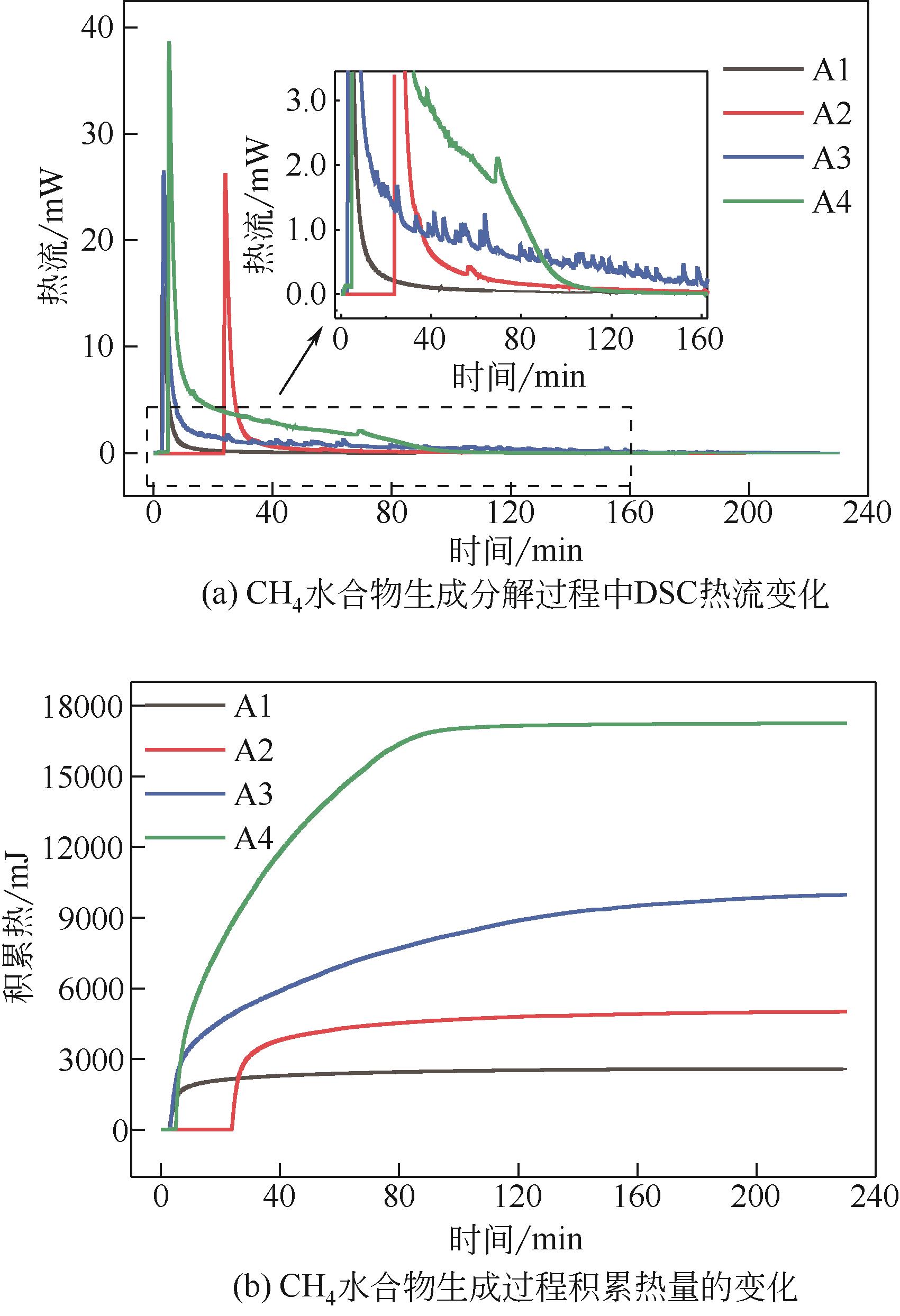
| 1 |
陈光进, 孙长宇, 马庆兰. 气体水合物科学与技术[M]. 北京:化学工业出版社, 2008.
|
|
CHEN G J, SUN C Y, MA Q L. Gas hydrate science and technology [M]. Beijing: Chemical Industry Press, 2008.
|
| 2 |
BOSWELL R, COLLETT T S. Current perspectives on gas hydrate resources[J]. Energy & Environmental Science, 2011, 4(4): 1206-1215.
|
| 3 |
SUN Z F, LI N, JIA S, et al. A novel method to enhance methane hydrate exploitation efficiency via forming impermeable overlying CO2 hydrate cap[J]. Applied Energy, 2019, 240: 842-850.
|
| 4 |
YANG X, SUN C Y, SU K H, et al. A three-dimensional study on the formation and dissociation of methane hydrate in porous sediment by depressurization[J]. Energy Conversion and Management, 2012, 56: 1-7.
|
| 5 |
YANG X, SUN C Y, YUAN Q, et al. Experimental study on gas production from methane hydrate-bearing sand by hot-water cyclic injection[J]. Energy & Fuels, 2010, 24(11): 5912-5920.
|
| 6 |
LI B, LIU S D, LIANG Y P, et al. The use of electrical heating for the enhancement of gas recovery from methane hydrate in porous media[J]. Applied Energy, 2018, 227: 694-702.
|
| 7 |
YUAN Q, SUN C Y, YANG X, et al. Gas production from methane-hydrate-bearing sands by ethylene glycol injection using a three-dimensional reactor[J]. Energy & Fuels, 2011, 25(7): 3108-3115.
|
| 8 |
SUN Y F, ZHONG J R, LI W Z, et al. Methane recovery from hydrate-bearing sediments by the combination of ethylene glycol injection and depressurization[J]. Energy & Fuels, 2018, 32(7): 7585-7594.
|
| 9 |
SUN Y F, ZHONG J R, LI R, et al. Natural gas hydrate exploitation by CO2/H2 continuous injection-production mode[J]. Applied Energy, 2018, 226: 10-21.
|
| 10 |
XIE Y, ZHU Y J, ZHENG T, et al. Replacement in CH4-CO2 hydrate below freezing point based on abnormal self-preservation differences of CH4 hydrate[J]. Chemical Engineering Journal, 2021, 403: 126283.
|
| 11 |
XIE Y, LI R, WANG X H, et al. Review on the accumulation behavior of natural gas hydrates in porous sediments[J]. Journal of Natural Gas Science and Engineering, 2020, 83: 103520.
|
| 12 |
HOLLAND M E, SCHULTHEISS P J, ROBERTS J A. Gas hydrate saturation and morphology from analysis of pressure cores acquired in the Bay of Bengal during expedition NGHP-02, offshore India[J]. Marine and Petroleum Geology, 2019, 108: 407-423.
|
| 13 |
SU K H, SUN C Y, DANDEKAR A, et al. Experimental investigation of hydrate accumulation distribution in gas seeping system using a large scale three-dimensional simulation device[J]. Chemical Engineering Science, 2012, 82: 246-259.
|
| 14 |
LI F G, SUN C Y, ZHANG Q, et al. Laboratory measurements of the effects of methane/tetrahydrofuran concentration and grain size on the P-wave velocity of hydrate-bearing sand[J]. Energy & Fuels, 2011, 25(5): 2076-2082.
|
| 15 |
LINGA P, HALIGVA C, NAM S C, et al. Gas hydrate formation in a variable volume bed of silica sand particles[J]. Energy & Fuels, 2009, 23(11): 5496-5507.
|
| 16 |
BAGHERZADEH S A, MOUDRAKOVSKI I L, RIPMEESTER J A, et al. Magnetic resonance imaging of gas hydrate formation in a bed of silica sand particles[J]. Energy & Fuels, 2011, 25(7): 3083-3092.
|
| 17 |
王英梅, 董世强, 展静, 等. 石英砂粒径大小对甲烷水合物形成及分布的影响[J]. 化工进展, 2020, 39(8): 3049-3056.
|
|
WANG Yingmei, DONG Shiqiang, ZHAN Jing, et al. Effect of quartz sand particle size on the formation and distribution of methane hydrate[J]. Chemical Industry and Engineering Progress, 2020, 39(8): 3049-3056.
|
| 18 |
XIE Y, ZHENG T, ZHONG J R, et al. Experimental research on self-preservation effect of methane hydrate in porous sediments[J]. Applied Energy, 2020, 268: 115008.
|
| 19 |
LIU H, ZHAN S Y, GUO P, et al. Understanding the characteristic of methane hydrate equilibrium in materials and its potential application[J]. Chemical Engineering Journal, 2018, 349: 775-781.
|
| 20 |
FITZGERALD G C, CASTALDI M J, SCHICKS J M. Methane hydrate formation and thermal based dissociation behavior in silica glass bead porous media[J]. Industrial & Engineering Chemistry Research, 2014, 53(16): 6840-6854.
|
| 21 |
LI N, SUN Z F, SUN C Y, et al. Simulating natural hydrate formation and accumulation in sediments from dissolved methane using a large three-dimensional simulator[J]. Fuel, 2018, 216: 612-620.
|
| 22 |
CHONG Z R, YANG M J, KHOO B C, et al. Size effect of porous media on methane hydrate formation and dissociation in an excess gas environment[J]. Industrial & Engineering Chemistry Research, 2016, 55(29): 7981-7991.
|
| 23 |
GUPTA A, LACHANCE J, SLOAN E D, et al. Measurements of methane hydrate heat of dissociation using high pressure differential scanning calorimetry[J]. Chemical Engineering Science, 2008, 63(24): 5848-5853.
|
| 24 |
STERN L A, KIRBY S H, DURHAM W B. Peculiarities of methane clathrate hydrate formation and solid-state deformation, including possible superheating of water ice[J]. Science, 1996, 273(5283): 1843-1848.
|
| 25 |
MOHAMMADI A H, AFZAL W, RICHON D. Gas hydrates of methane, ethane, propane, and carbon dioxide in the presence of single NaCl, KCl, and CaCl2 aqueous solutions: experimental measurements and predictions of dissociation conditions[J]. The Journal of Chemical Thermodynamics, 2008, 40(12): 1693-1697.
|
 ), 谢炎(
), 谢炎( ), 钟瑾荣, 孙长宇(
), 钟瑾荣, 孙长宇( ), 陈光进, 王晓辉
), 陈光进, 王晓辉
 ), XIE Yan(
), XIE Yan( ), ZHONG Jinrong, SUN Changyu(
), ZHONG Jinrong, SUN Changyu( ), CHEN Guangjin, WANG Xiaohui
), CHEN Guangjin, WANG Xiaohui