化工进展 ›› 2019, Vol. 38 ›› Issue (06): 2870-2879.DOI: 10.16085/j.issn.1000-6613.2018-1866
氧化石墨烯-TiO2复合材料对三种染料的吸附动力学及光催化性能
- 中北大学超重力化工过程山西省重点实验室,山西 太原 030051
-
收稿日期:2018-09-17出版日期:2019-06-05发布日期:2019-06-05 -
作者简介:张巧玲(1965—),女,教授,硕士生导师,研究方向为二氧化钛的改性。E-mail:<email>zhangqiaoling@nuc.edu.cn</email>。
Adsorption kinetics and photocatalytic activity of grapheneoxide-TiO2 composites for three dyes
Qiaoling ZHANG( ),Zhao QIN,Youzhi LIU,Yanting SHI,Jingwen ZHANG,Guangping ZENG
),Zhao QIN,Youzhi LIU,Yanting SHI,Jingwen ZHANG,Guangping ZENG
- Shanxi Province Key Laboratory of Higee-Oriented Chemical Engineering, North University of China, Taiyuan 030051, Shanxi, China
-
Received:2018-09-17Online:2019-06-05Published:2019-06-05
摘要:
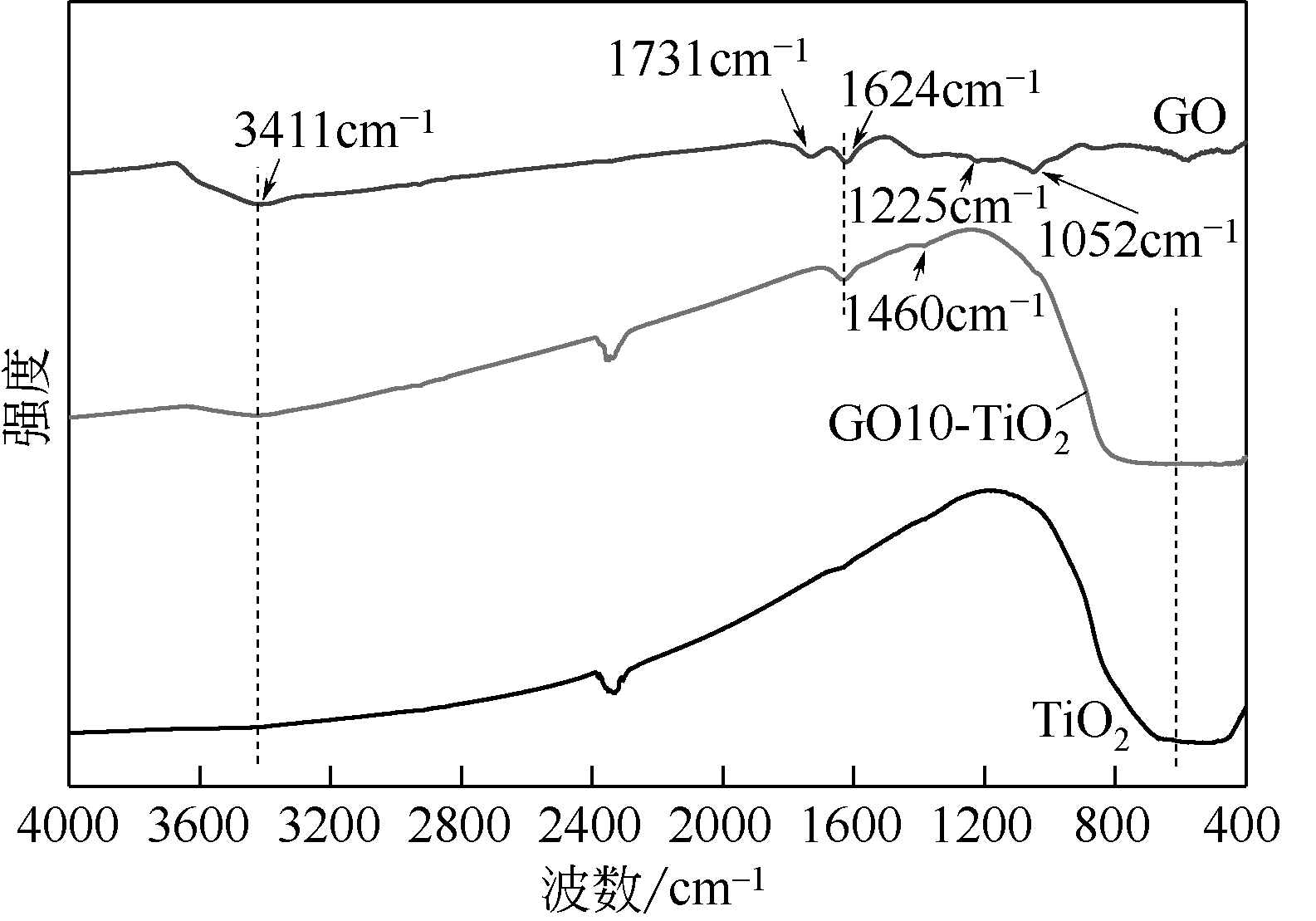
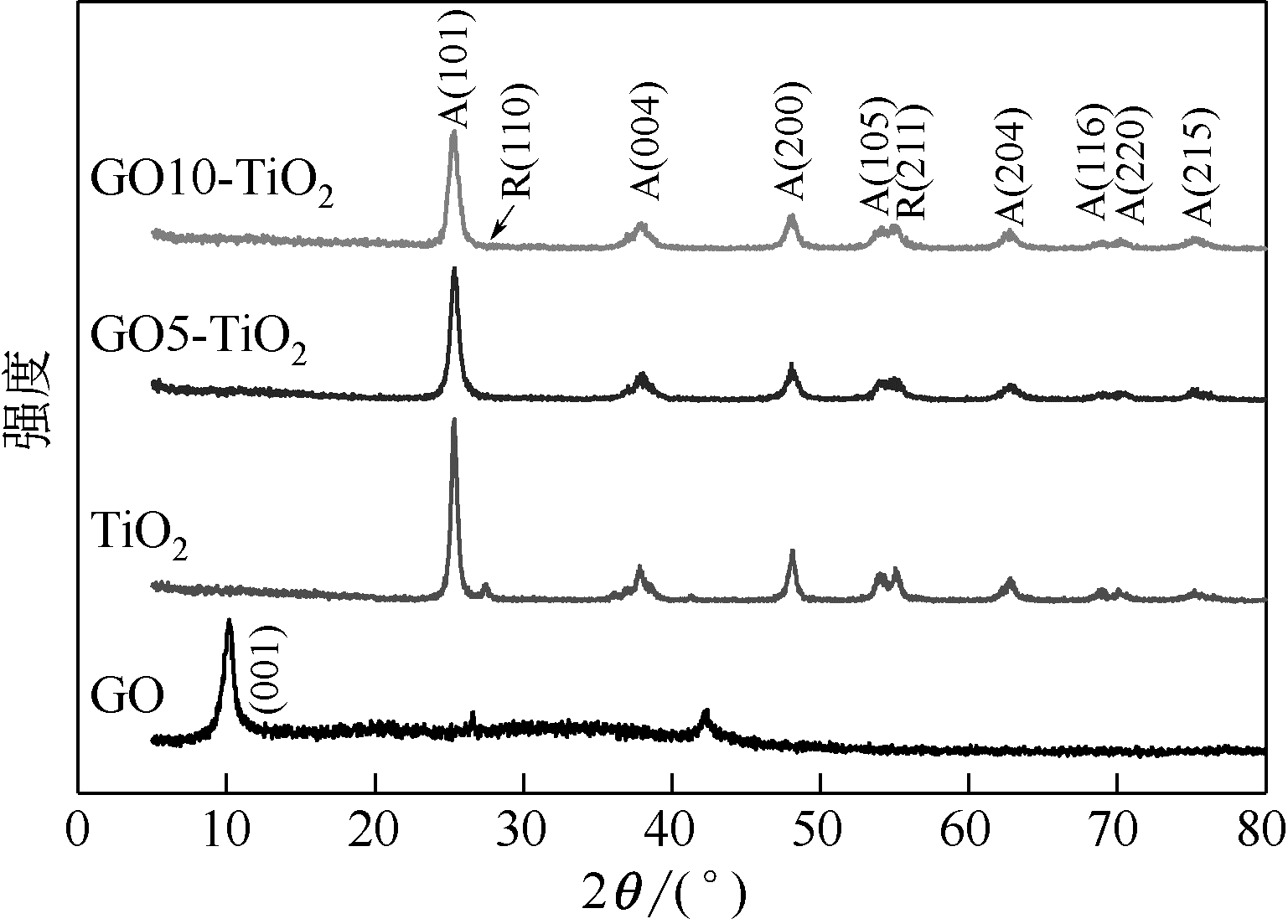
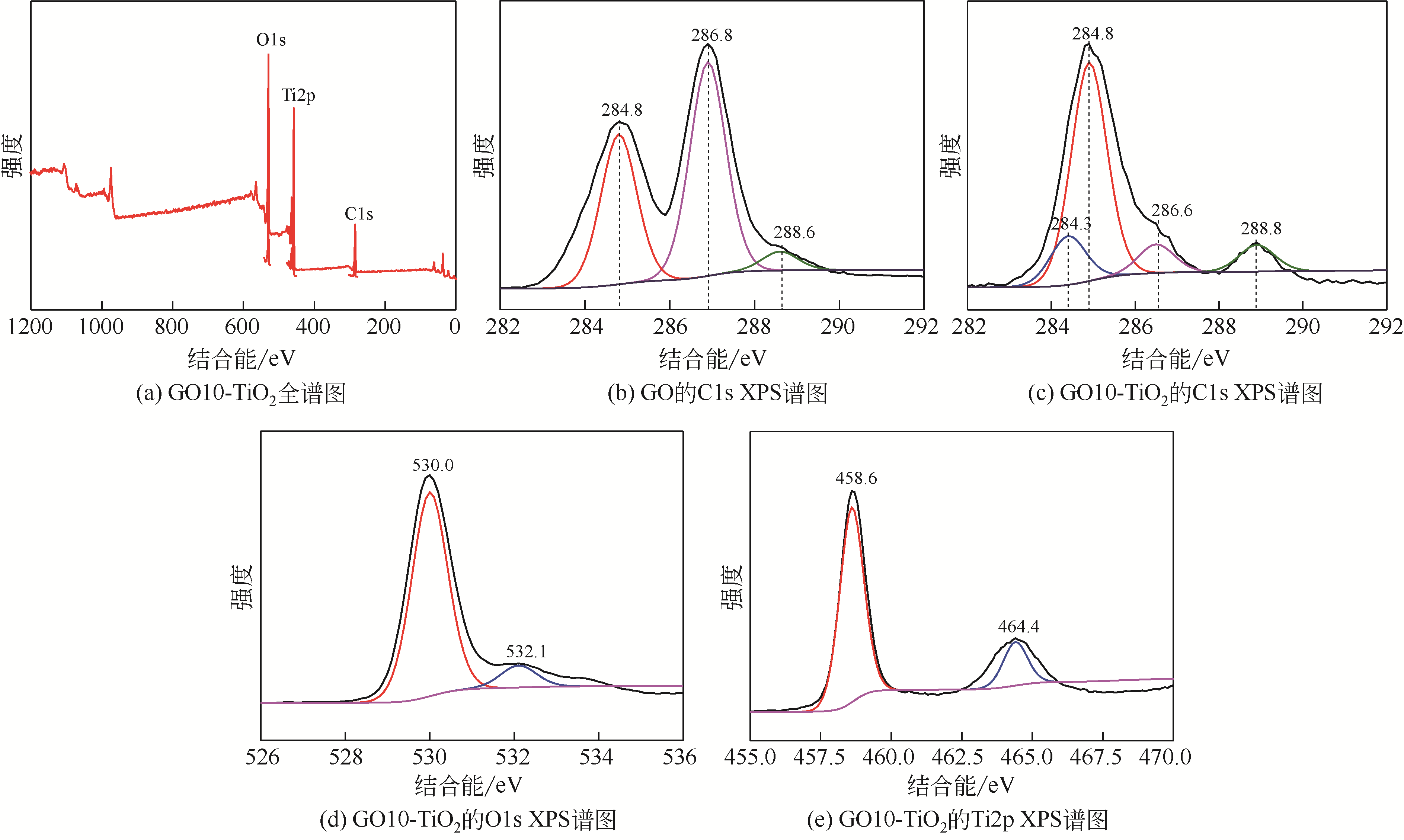
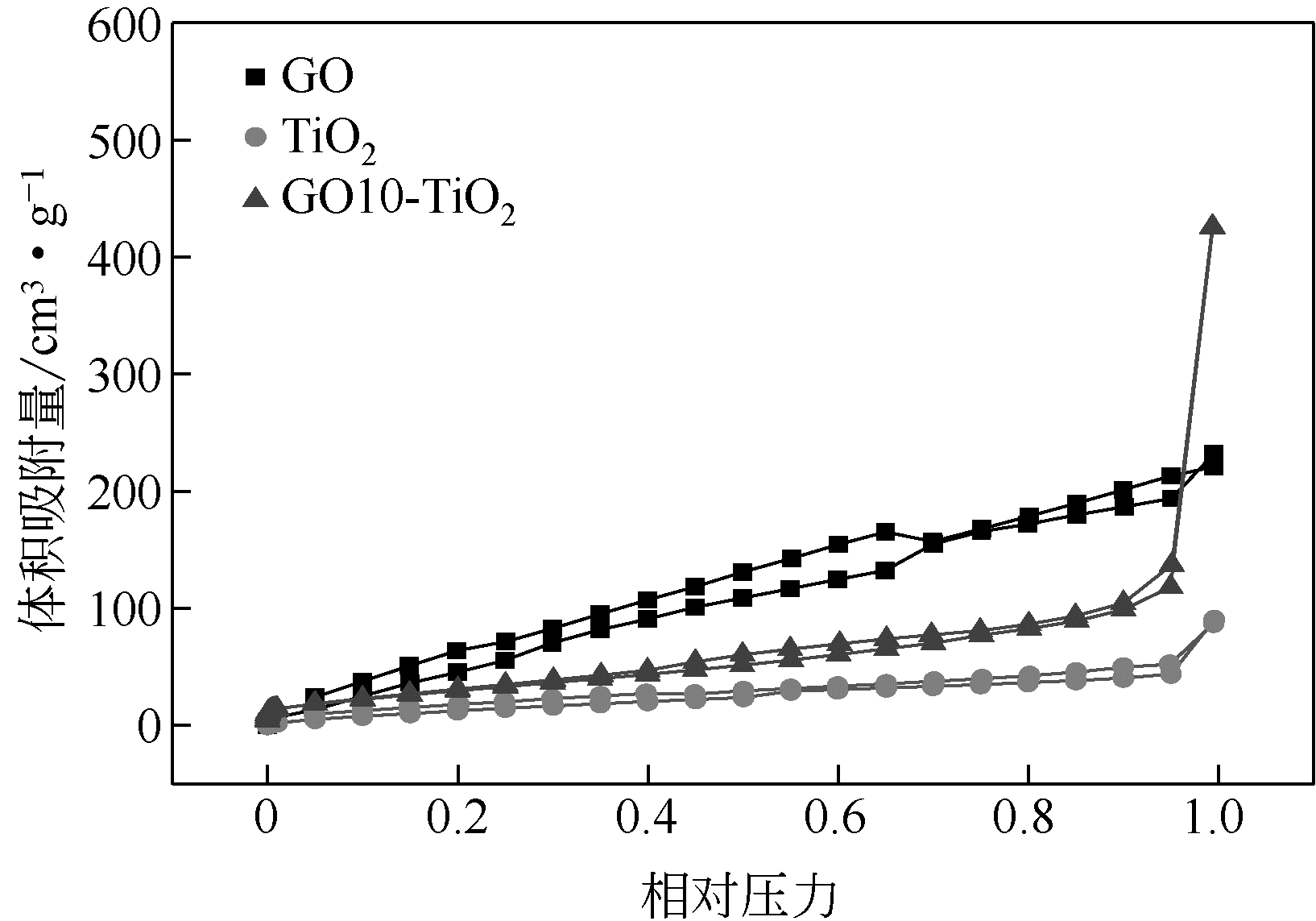
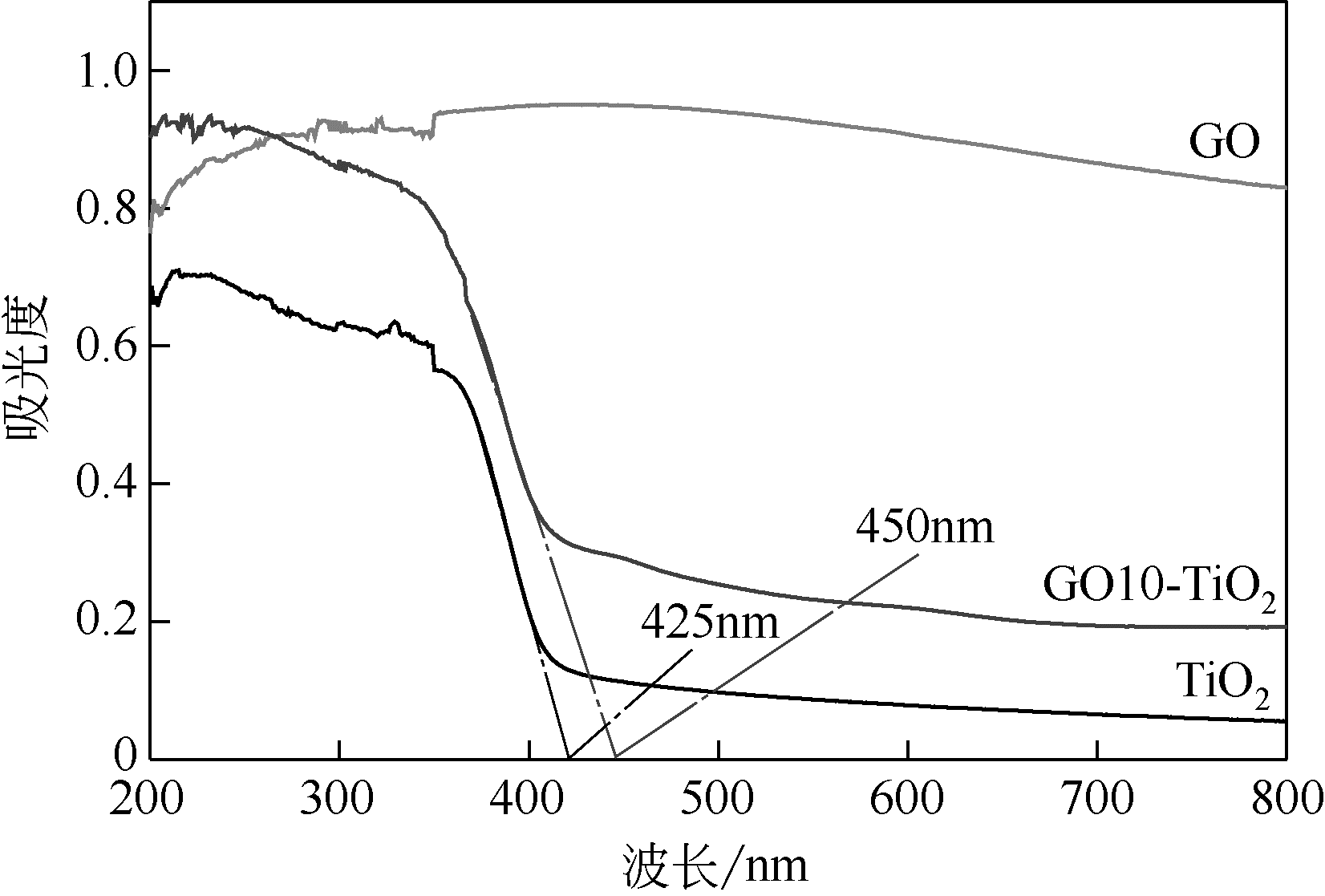
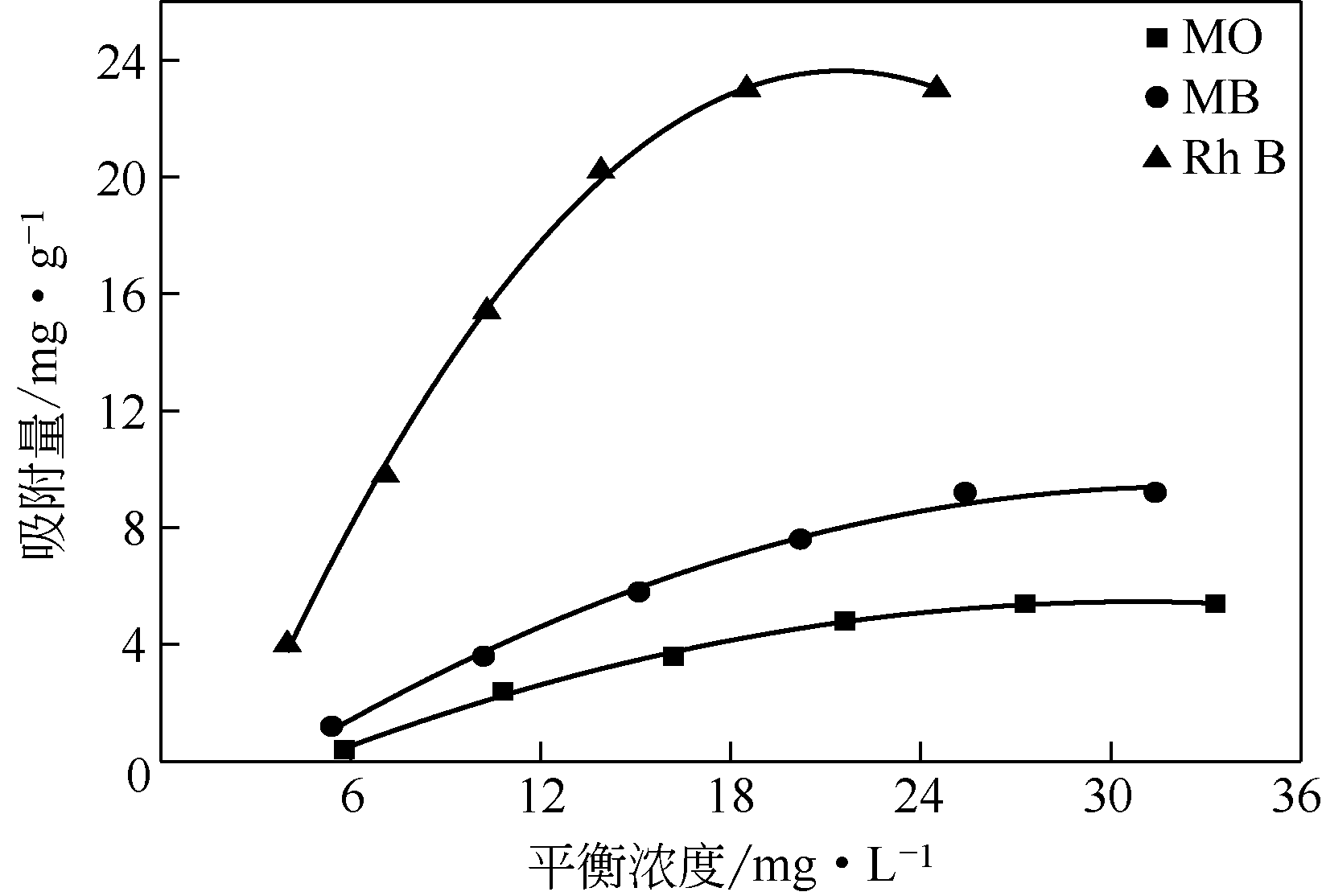
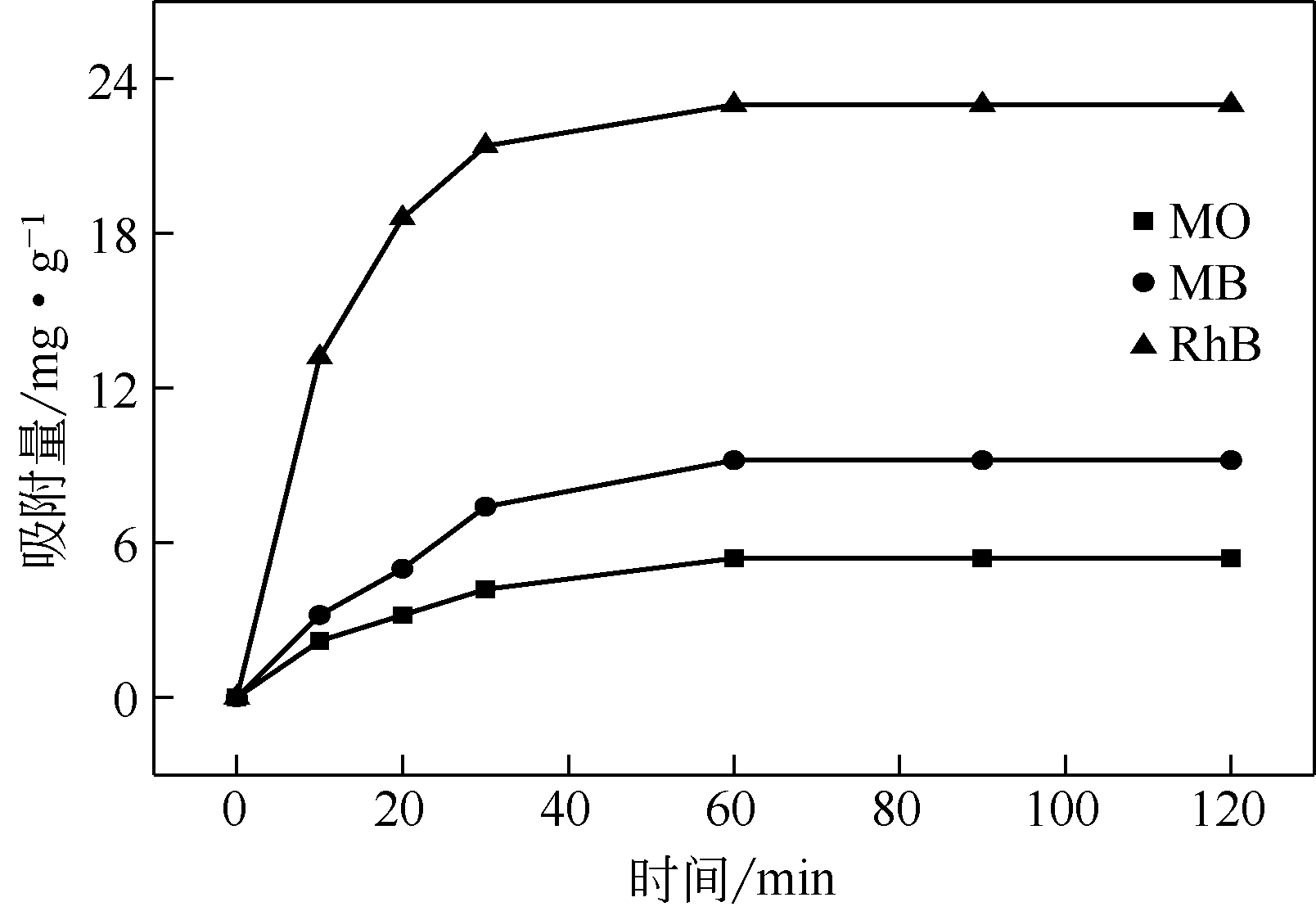
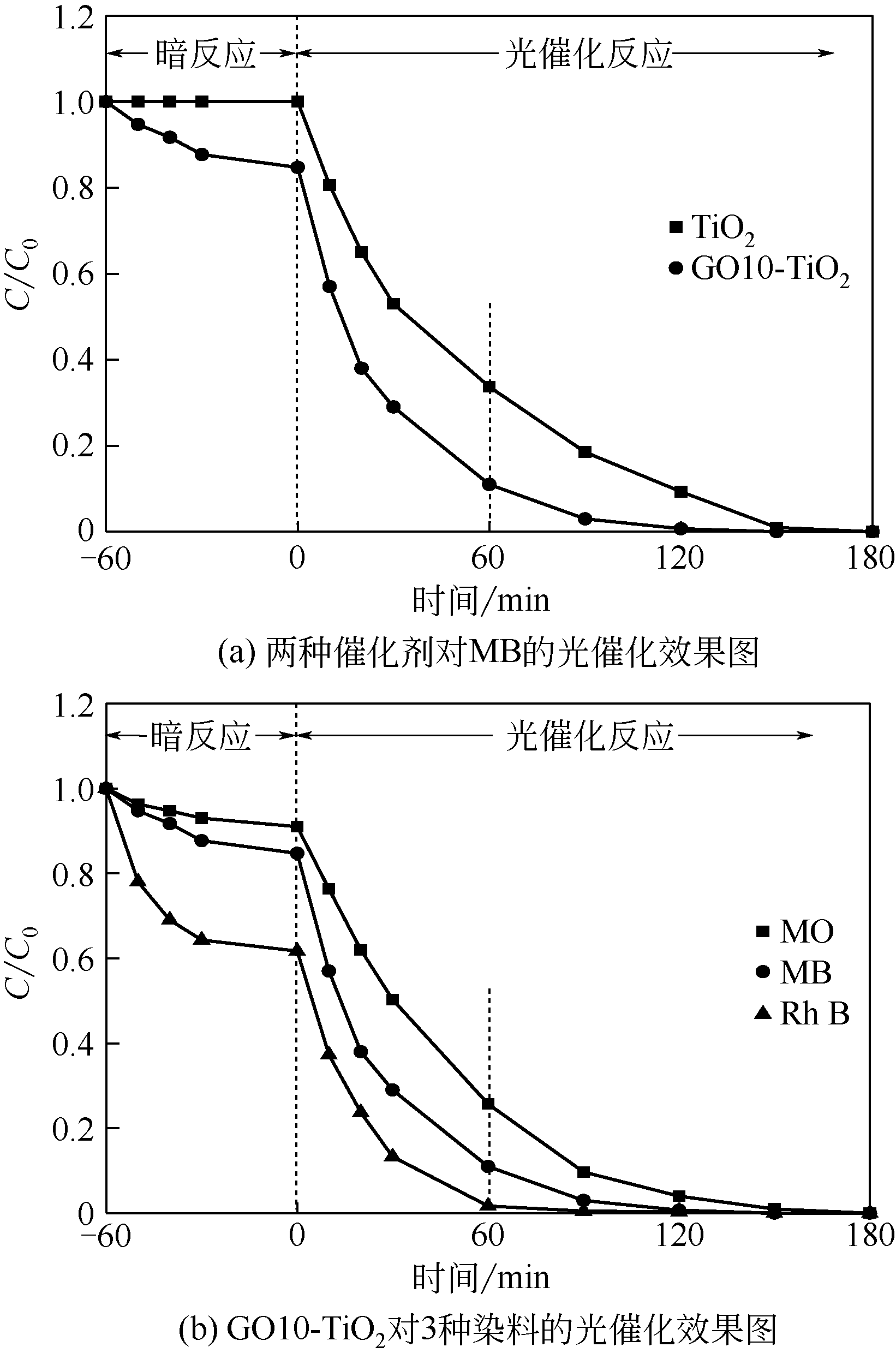
通过在氧化石墨烯分散溶液中水解钛酸丁酯成功制备氧化石墨烯-TiO2复合材料(GO-TiO2),采用傅里叶变换红外光谱(FTIR)、X射线衍射(XRD)、扫描电子显微镜(SEM)、X射线光电子能谱(XPS)、全自动比表面及孔径分析仪(BET)和紫外-可见漫反射光谱(UV-vis DRS)等对样品进行了表征。研究了GO10-TiO2对亚甲基蓝(MB)、甲基橙(MO)和罗丹明B(RhB)3种染料的吸附动力学和光催化性能。结果表明:TiO2颗粒均匀地附着在GO片层表面;GO10-TiO2对3种染料的吸附过程为多层吸附,吸附动力学符合拟二级动力学模型;在25℃条件下GO10-TiO2对废水中MB、MO和Rh B的吸附因共轭结构、极性等的差异而呈现选择性吸附,吸附容量分别为9.2mg/g、5.4mg/g和23.0mg/g。对3种染料废水的光催化降解效果与吸附性能相关联,吸附容量越大降解效率越高,光催化反应60min时,MB、MO和Rh B降解率分别为89%、75%和98%。
中图分类号:
引用本文
张巧玲, 秦钊, 刘有智, 师艳婷, 张竞文, 曾广平. 氧化石墨烯-TiO2复合材料对三种染料的吸附动力学及光催化性能[J]. 化工进展, 2019, 38(06): 2870-2879.
Qiaoling ZHANG, Zhao QIN, Youzhi LIU, Yanting SHI, Jingwen ZHANG, Guangping ZENG. Adsorption kinetics and photocatalytic activity of grapheneoxide-TiO2 composites for three dyes[J]. Chemical Industry and Engineering Progress, 2019, 38(06): 2870-2879.
| 2θ/(°) | 晶面 | 2θ/(°) | 晶面 |
|---|---|---|---|
| 25 | (101) | 62 | (204) |
| 38 | (004) | 69 | (116) |
| 48 | (200) | 70 | (220) |
| 53 | (105) | 75 | (215) |
表1 锐钛矿相的晶面所对应的2θ角
| 2θ/(°) | 晶面 | 2θ/(°) | 晶面 |
|---|---|---|---|
| 25 | (101) | 62 | (204) |
| 38 | (004) | 69 | (116) |
| 48 | (200) | 70 | (220) |
| 53 | (105) | 75 | (215) |
| 材料 | 比表面积/m2·g-1 |
|---|---|
| GO | 339.0 |
| TiO2 | 53.9 |
| GO1-TiO2 | 63.8 |
| GO5-TiO2 | 89.7 |
| GO10-TiO2 | 109.7 |
| GO15-TiO2 | 121.1 |
| GO20-TiO2 | 134.6 |
表3 不同材料的比表面积
| 材料 | 比表面积/m2·g-1 |
|---|---|
| GO | 339.0 |
| TiO2 | 53.9 |
| GO1-TiO2 | 63.8 |
| GO5-TiO2 | 89.7 |
| GO10-TiO2 | 109.7 |
| GO15-TiO2 | 121.1 |
| GO20-TiO2 | 134.6 |
| 复合材料 | 吸附率/% | 60min总降解率/% |
|---|---|---|
| TiO2 | 0 | 52.4 |
| GO1-TiO2 | 6.3 | 85.3 |
| GO5-TiO2 | 8.0 | 87.3 |
| GO10-TiO2 | 15.3 | 94.0 |
| GO15-TiO2 | 16.7 | 65.0 |
| GO20-TiO2 | 17.0 | 52.7 |
表4 不同复合材料对MB的吸附率和光催化降解率
| 复合材料 | 吸附率/% | 60min总降解率/% |
|---|---|---|
| TiO2 | 0 | 52.4 |
| GO1-TiO2 | 6.3 | 85.3 |
| GO5-TiO2 | 8.0 | 87.3 |
| GO10-TiO2 | 15.3 | 94.0 |
| GO15-TiO2 | 16.7 | 65.0 |
| GO20-TiO2 | 17.0 | 52.7 |
| 染料 | 结构式 | 相对分子质量 | 极性汉森参数δ p |
|---|---|---|---|
| MB | | 319 | 2.904 |
| MO | | 327 | 3.113 |
| Rh B | | 479 | 4.298 |
表5 3种染料的极性汉森参数
| 染料 | 结构式 | 相对分子质量 | 极性汉森参数δ p |
|---|---|---|---|
| MB | | 319 | 2.904 |
| MO | | 327 | 3.113 |
| Rh B | | 479 | 4.298 |
| 染料 | q m,exp /mg·g-1 | Langmuir 模型 | Freundlich 模型 | ||||
|---|---|---|---|---|---|---|---|
| q m,cal/mg·g-1 | K L/L·mg-1 | R 2 | K F | n | R 2 | ||
| MB | 9.2 | 10.10 | 0.020 | 0.734 | 0.114 | 0.726 | 0.982 |
| MO | 5.4 | 2.53 | 0.027 | 0.701 | 0.023 | 0.603 | 0.994 |
| Rh B | 23.0 | 26,31 | 0.038 | 0.763 | 0.902 | 0.859 | 0.993 |
表6 GO10-TiO2催化剂对3种染料的吸附等温线模型的相关参数
| 染料 | q m,exp /mg·g-1 | Langmuir 模型 | Freundlich 模型 | ||||
|---|---|---|---|---|---|---|---|
| q m,cal/mg·g-1 | K L/L·mg-1 | R 2 | K F | n | R 2 | ||
| MB | 9.2 | 10.10 | 0.020 | 0.734 | 0.114 | 0.726 | 0.982 |
| MO | 5.4 | 2.53 | 0.027 | 0.701 | 0.023 | 0.603 | 0.994 |
| Rh B | 23.0 | 26,31 | 0.038 | 0.763 | 0.902 | 0.859 | 0.993 |
| 染料 | q e,exp /mg·g-1 | 拟一级动力学 | 拟二级动力学 | 颗粒内扩散模型 | |||||
|---|---|---|---|---|---|---|---|---|---|
| q e,cal /mg·g-1 | k 1 /min-1 | R 2 | q e,cal /mg·g-1 | k 2 /mg·g-1·min-1 | R 2 | ki, 1 /mg·g-1·min-1/2 | ki, 2 /mg·g-1·min-1/2 | ||
| MB | 9.203 | 9.936 | 0.053 | 0.937 | 9.379 | 11.43×10-3 | 0.991 | 1.331 | 0.083 |
| MO | 5.421 | 5.708 | 0.049 | 0.988 | 5.408 | 34.19×10-3 | 0.990 | 0.701 | 0.056 |
| Rh B | 23.123 | 26.316 | 0.088 | 0.986 | 24.456 | 1.672×10-3 | 0.997 | 2.042 | 0.102 |
表7 GO10-TiO2催化剂对3种有机物的吸附动力学模型相关参数
| 染料 | q e,exp /mg·g-1 | 拟一级动力学 | 拟二级动力学 | 颗粒内扩散模型 | |||||
|---|---|---|---|---|---|---|---|---|---|
| q e,cal /mg·g-1 | k 1 /min-1 | R 2 | q e,cal /mg·g-1 | k 2 /mg·g-1·min-1 | R 2 | ki, 1 /mg·g-1·min-1/2 | ki, 2 /mg·g-1·min-1/2 | ||
| MB | 9.203 | 9.936 | 0.053 | 0.937 | 9.379 | 11.43×10-3 | 0.991 | 1.331 | 0.083 |
| MO | 5.421 | 5.708 | 0.049 | 0.988 | 5.408 | 34.19×10-3 | 0.990 | 0.701 | 0.056 |
| Rh B | 23.123 | 26.316 | 0.088 | 0.986 | 24.456 | 1.672×10-3 | 0.997 | 2.042 | 0.102 |
| 1 | WANG Z , MA W, CHEN C , et al . Light-assisted decomposition of dyes over iron-bearing soil clays in the presence of H2O2 [J]. Journal of Hazardous Materials, 2009,168 (2): 1246-1252. |
| 2 | 张志军,胡涓,陈整生,等 . 纳米二氧化钛复合石墨烯催化剂的制备及处理染料废水[J]. 环境工程学报, 2014, 8(7): 2875-2879. |
| ZHANG Zhijun , HU Juan , CHEN Zhengsheng ,et al . Preparation of nano-TiO2 composite graphene catalyst and treatment of dye wastewater[J]. Journal of Environmental Engineering, 2014, 8(7): 2875-2879 . | |
| 3 | JUANG R S , CHEN C H . Comparative study on photocatalytic degradation of methomyl and parathion over UV-irradiated TiO2 particles in aqueous solutions[J]. Journal of the Taiwan Institute of Chemical Engineers, 2014, 45(3): 989-995. |
| 4 | MATTSSON A , STERLUND L . Adsorption and photoinduced decomposition of acetone and acetic acid on anatase, brookite, and rutile TiO2 nanoparticles[J]. The Journal of Physical Chemistry C, 2010, 114(33): 14121-14132. |
| 5 | 李翠霞,金海泽,杨志忠,等 . 介孔RGO/TiO2复合光催化材料的制备及光催化性能[J]. 无机材料学报, 2017, 32(4): 357-364. |
| LI Cuixia , JIN Haize , YANG Zhizhong ,et al . Preparation and photocatalytic activity of mesoporous RGO/TiO2 composite photocatalytic materials[J]. Journal of Inorganic Materials, 2017, 32(4): 357-364. | |
| 6 | YANGAB L , WANGA F , HAKKIB A , et al . The influence of zeolites fly ash bead/TiO2 composite material surface morphologies on their adsorption and photocatalytic performance[J]. Applied Surface Science, 2017, 329: 687-696. |
| 7 | MUTHIRULAN P , DEVI C N , SUNDARAM M M . Synchronous role of coupled adsorption and photocatalytic degradation on CAC-TiO2 composite generating excellent mineralization of alizarin cyanine green dye in aqueous solution[J]. Arabian Journal of Chemistry, 2017, 10(1): S1477-S1483. |
| 8 | ZHOUAB J , HAOA B , WANGA L , et al . Preparation and characterization of nano-TiO2/chitosan/poly(N-isopropylacrylamide) composite hydrogel and its application for removal of ionic dyes[J]. Separation and Purification Technology, 2017,176: 193-199. |
| 9 | ZHANG J , LI L , LI Y , et al . Microwave-assisted synthesis of hierarchical mesoporous nano-TiO2/cellulose composites for rapid adsorption of Pb2+ [J]. Chemical Engineering Journal, 2017,313: 1132-1141. |
| 10 | BAI H , ZHOU J , ZHANG H , et al . Enhanced adsorbability and photocatalytic activity of TiO2-graphene composite for polycyclic aromatic hydrocarbons removal in aqueous phase[J]. Colloids and Surfaces B-Biointerfaces, 2017, 150: 68-77. |
| 11 | WANG D , LI X , CHEN J , et al . Enhanced photoelectrocatalytic activity of reduced graphene oxide/TiO2 composite films for dye degradation[J]. Chemical Engineering Journal, 2012, 198/199(4): 547-554. |
| 12 | YANG Wenrong , K R RATINAC , RINGER S P , et al . Carbon nanomaterials in biosensors: should you use nanotubes or graphene[J]. Angewandte Chemie International Edition, 2010, 41(24): 2114-2138. |
| 13 | DONDONIR A . The emergence of thiolene coupling as a click process for materials and bioorganic chemistry[J]. Angewandte Chemie, 2008, 47(47): 8995-8997. |
| 14 | PUMERA M , AMBROSI A , BONANNI A , et al . Graphene for electrochemical sensing and biosensing[J]. Trends in Analytical Chemistry, 2010, 29(9): 954-965. |
| 15 | DREYER D R , PARK S , BIELAWSKI C W , et al . The chemistry of graphene oxide[J]. Chemical Society Reviews, 2010, 43(15) : 5288. |
| 16 | DIKIN D A , STANKOVICH S , ZIMNEY E J , et al . Preparation and characterization of graphene oxide paper[J]. Nature, 2007, 448(7152): 457-460. |
| 17 | DONG Z , WANG D , LIU X , et al . Bio-inspired surface-functionalization of graphene oxide for the adsorption of organic dyes and heavy metal ions with a superhigh capacity[J]. Journal of Materials Chemistry A, 2014, 2(14): 5034-5040. |
| 18 | BISSESSUR R , SCULLY S F . Intercalation of solid polymer electrolytes into graphite oxide[J]. Solid State Ionics, 2007, 178(11): 877-882. |
| 19 | ZHANG H , LV X , LI Y , et al . P25-graphene composite as a high performance photocatalyst[J]. ACS Nano, 2010, 4(1): 380-386. |
| 20 | PERERA S D , MARIANO R G , VU K, et al . Hydrothermal synthesis of graphene-TiO2 nanotube composites with enhanced photocatalytic activity[J]. ACS Catalysis, 2012, 2(6): 949-956. |
| 21 | ZHANG L , LIU J , TANG C , et al . Palygorskite and SnO2–TiO2 for the photodegradation of phenol[J]. Applied Clay Science, 2011, 51(1/2): 68-73. |
| 22 | LI L , DUAN H , WANG X , et al . Adsorption property of Cr(VI) on magnetic mesoporous titanium dioxide–graphene oxide core–shell microspheres[J]. New Journal of Chemistry, 2014, 38(12): 6008-6016. |
| 23 | LEI Z , JO S B, SHU Y , et al . Rhodamine B degradation and reactive oxygen species generation by a ZnSe-graphene/TiO2 sonocatalyst[J]. Chinese Journal of Catalysis, 2014, 35(11):1825-1832. |
| 24 | MURUGAN R , BABU V J , KHIN M M , et al . Synthesis and photocatalytic applications of flower shaped electrospun ZnO-TiO2 mesostructures[J]. Materials Letters, 2013, 97(2): 47-51. |
| 25 | CAOA P , WANGA L , XUA Y, et al . Facile hydrothermal synthesis of mesoporous nickel oxide/reduced graphene oxide composites for high performance electrochemical supercapacitor[J]. Electrochimica Acta, 2015, 157: 359-368. |
| 26 | LIU R , ZHENG Z , SPURGEON J , et al . Enhanced photoelectrochemical water-splitting performance of semiconductors by surface passivation layers[J]. Energy and Environmental Science, 2014, 7(8): 2504-2517. |
| 27 | HANSEN C M . Hansen solubility parameters: a user’s handbook[M]. Boca Raton: CRC Press Inc. , 2007:493-505. |
| 28 | DESLANDES N , BELLENGER V , JAFFIOL F , et al . Solubility parameter of a polyester composite material[J]. Journal of Applied Polymer Science, 2015, 69(13): 2663-2671. |
| 29 | SCHRODERA J H , DOROSHENKOAD M , PIMERA D , et al . Interfacial stabilization by soft janus nanoparticles[J]. Polymer, 2016, 106: 208-217. |
| 30 | 刘桂芳 . 常用吸附材料在水处理中的应用[M].北京:化学工业出版社, 2016:71-84. |
| LIU Guifang . Application of common adsorbent materials in water treatment[M]. Beijing: Chemical Industry Press, 2016:71-84. | |
| 31 | YUJUAN H , KAN Z , RONG D , et al . Enhanced adsorption of methyl orange onto self-assembled hydrogel with anatase titania nanotube and graphene[J]. Journal of Donghua University:English Edition, 2017, 34(2): 262-268. |
| [1] | 许春树, 姚庆达, 梁永贤, 周华龙. 氧化石墨烯/碳纳米管对几种典型高分子材料的性能影响[J]. 化工进展, 2023, 42(6): 3012-3028. |
| [2] | 张浩月, 李春丽, 徐博, 李筱贺, 仝铃, 邱广明. 分段取样法研究改进Hummers法制备GO结构特性及其机理[J]. 化工进展, 2023, 42(5): 2606-2615. |
| [3] | 何阳, 李思盈, 李传强, 袁小亚, 郑旭煦. 热还原氧化石墨烯/环氧树脂复合涂层的防腐性能[J]. 化工进展, 2023, 42(4): 1983-1994. |
| [4] | 孔倩, 孙巾超, 葛佳琪, 张鹏, 马艳龙, 刘百军. 沉淀剂对NiW/TiO2-ASA催化剂加氢裂化性能的影响[J]. 化工进展, 2023, 42(1): 265-271. |
| [5] | 袁权, 李海红, 刘浩杰. HNO3改性活性炭对不同价态离子的电吸附规律[J]. 化工进展, 2022, 41(9): 4986-4994. |
| [6] | 熊剑, 夏柳芬, 虞雷, 费安杰, 徐楚, 陈盛亚, 江国栋. 氧化石墨烯复合TiO2-B薄膜的制备及其电致变色性能[J]. 化工进展, 2022, 41(7): 3794-3800. |
| [7] | 陈勇, 程宁, 杨育兵, 卢凯玲, 罗应, 易慧. 氧化石墨烯插层膨润土复合材料高效吸附碱性紫3染料[J]. 化工进展, 2022, 41(6): 3324-3332. |
| [8] | 杨洪海, 张苗, 刘利伟, 周屹, 沈俊杰, 施伟刚, 尹勇. 氧化石墨烯/水脉动热管传热强化及性能预测[J]. 化工进展, 2022, 41(4): 1725-1734. |
| [9] | 毛梦雷, 孙丹阳, 孟子晖, 刘文芳. 氧化石墨烯和过渡金属碳/氮化合物固定化酶[J]. 化工进展, 2022, 41(4): 1941-1955. |
| [10] | 戴绍铃, 于桢, 李逸航, 成少安. 多层纳米结构蓝色TiO2的电化学氧化性能和稳定性[J]. 化工进展, 2022, 41(2): 862-873. |
| [11] | 崔维怡, 丁国敏, 谭乃迪. 二氧化钛基催化剂催化氧化甲醛的研究进展[J]. 化工进展, 2022, 41(12): 6310-6318. |
| [12] | 胡楠, 陈林, 李会珍, 张思瑶, 张志军. 强化泡沫排液下浮选富集和回收工程纳米颗粒[J]. 化工进展, 2022, 41(1): 485-492. |
| [13] | 郑安达, 杨成功, 王冬娥, 田志坚. 水热合成rGO负载的MoS2催化剂及其催化蒽加氢性能[J]. 化工进展, 2022, 41(1): 244-252. |
| [14] | 孙通, 许东东, 宋民航, 靳星, 黄云. 火焰合成法制备TiO2的燃烧发生器研究进展[J]. 化工进展, 2022, 41(1): 17-29. |
| [15] | 杨晓芳, 魏铭, 孙力. 聚丙烯酰胺/碳量子点/氧化石墨烯复合水凝胶制备及其性能分析[J]. 化工进展, 2021, 40(S2): 301-308. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||