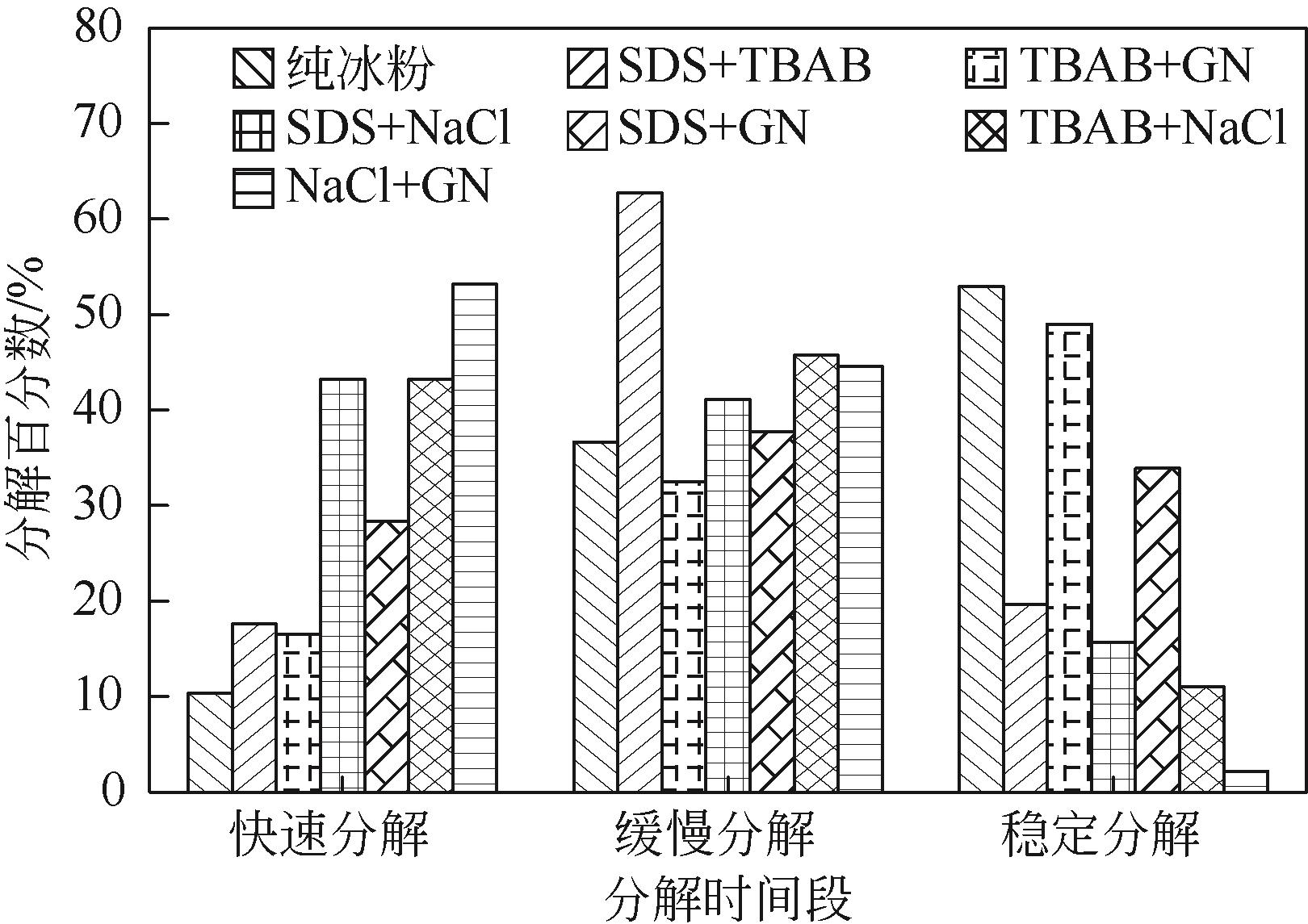
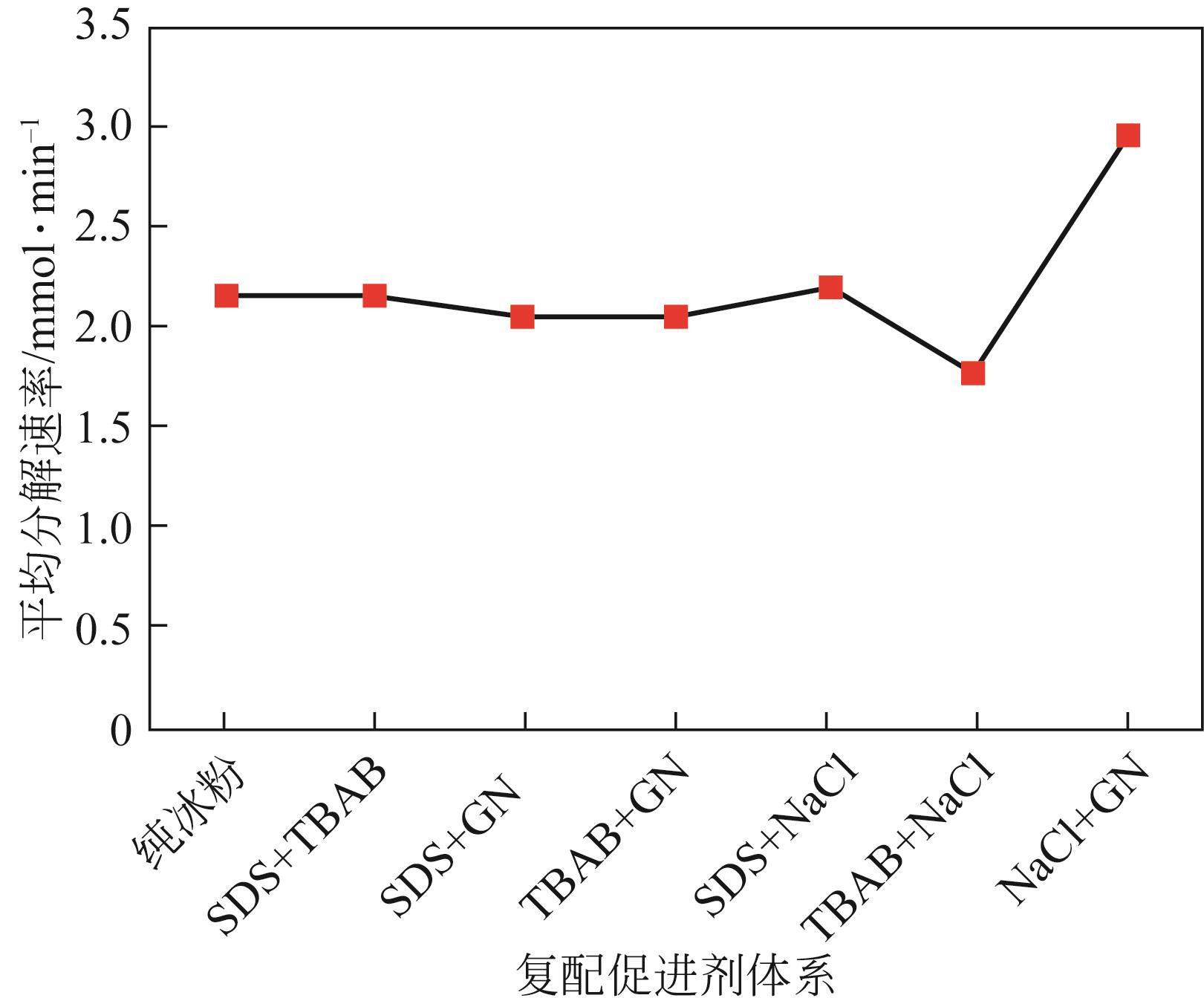
| 1 |
TAHERI Z, SHABANI M R, NAZARI K, et al. Natural gas transportation and storage by hydrate technology: iran case study[J]. Journal of Natural Gas Science and Engineering, 2014, 21(3): 846-849.
|
| 2 |
CASTELLANI B, FILIPPONI M, NICOLINI A, et al. Carbon dioxide capture using gas hydrate technology[J]. Journal of Energy and Power Engineering, 2013, 7(7): 883-889.
|
| 3 |
VELUSWAMY H P, BHATTACHARJEE G, LIAO J, et al. Macroscopic kinetic investigations on mixed natural gas hydrate formation for gas storage application[J]. Energy & Fuels, 2020, 34(2): 15257-15269.
|
| 4 |
YAMASAKI A, WAKATSUKI M, TENG H, et al. A new ocean disposal scenario for anthropogenic CO2: CO2 hydrate formation in a submerged crystallizer and its disposal[J]. Energy, 2000, 25(9): 85-96.
|
| 5 |
MATSUI H, JIA J, TSUJI T, et al. Microsecond simulation study on the replacement of methane in methane hydrate by carbon dioxide, nitrogen, and carbon dioxide–nitrogen mixtures[J]. Fuel, 2020, 263(8): 116-640.
|
| 6 |
XIE Y, ZHU Y J, ZHENG T, et al. Replacement in CH4-CO2 hydrate below freezing point based on abnormal self-preservation differences of CH4 hydrate[J]. Chemical Engineering Journal, 2021, 403(5): 126-283.
|
| 7 |
RYOU J E, AI-RAOUSH R I, AISHIBLI K, et al. Effects of soaking process on CH4-CO2 replacement efficiency for hydrate-bearing sediments[J]. Journal of Petroleum Science and Engineering, 2021, 196(8): 107-772.
|
| 8 |
CHOI W, LEE Y, MOK J, et al. Influence of competitive inclusion of CO2 and N2 on SⅡ hydrate–flue gas replacement for energy recovery and CO2 sequestration[J]. Environmental Science & Technology, 2020, 54(4): 7562-7569.
|
| 9 |
ERSHOV E D, YAKUSHEV V S. Experimental research on gas hydrate decomposition in frozen rocks[J]. Cold Regions Science and Technology, 1992, 20(2): 147-156.
|
| 10 |
GUDMUNDSSON J S, PARLAKTUNA M, LEVIK O I, et al. Laboratory for continuous production of natural gas hydrates[J]. Annals of the New York academy of Sciences, 2000, 912(4): 851-858.
|
| 11 |
孙腾民, 刘世奇, 汪涛. 中国二氧化碳地质封存潜力评价研究进展[J]. 煤炭科学技术, 2021, 49(11): 10-20.
|
|
SUN Tengmin, LIU Shiqi, WANG Tao. Research advances on evaluation of CO2 geological storage potential in China[J]. Coal Science and Technology, 2021, 49(11): 10-20.
|
| 12 |
LIN W, CHEN G J, SUN C Y, et al. Effect of surfactant on the formation and dissociation kinetic behavior of methane hydrate[J]. Chemical Engineering Science, 2004, 59(21): 4449-4455.
|
| 13 |
CHENG Chuanxiao, WANG Fan, ZHANG Jun, et al. Cyclic formation stability of 1,1,1,2-tetrafluoroethane hydrate in different SDS solution systems and dissociation characteristics using thermal stimulation combined with depressurization[J]. ACS Omega, 2019, 4(7): 11397-11407.
|
| 14 |
ZHANG H, XU J, LI Z, et al. Effect of electrolytes on the self-protection of natural gas hydrate decomposition[J]. Journal of Geophysics and Engineering, 2021, 18 (4): 482-491.
|
| 15 |
GE B B, LI X Y, ZHONG D L, et al. Investigation of natural gas storage and transportation by gas hydrate formation in the presence of bio-surfactant sulfonated lignin[J]. Energy, 2022, 244: 122665.
|
| 16 |
JIANG L L, LI A R, XU J F, et al. Effects of SDS and SDBS on CO2 hydrate formation, induction time, storage capacity and stability at 274.15K and 5.0 MPa[J]. ChemistrySelect, 2016, 1(7):6111-6114.
|
| 17 |
SUN Zhigao, JIAO Lijun, ZHAO Zhigui, et al. Phase equilibrium conditions of semi-calthrate hydrates of (tetra-n-butyl ammonium chloride + carbon dioxide)[J]. Journal of Chemical Thermodynamics, 2014, 75: 116-118.
|
| 18 |
FAKHARIAN H, GANJI H, FAR A N, et al. Potato starch as methane hydrate promoter[J]. Fuel, 2012, 94(8): 356-360.
|
| 19 |
ZENG Y, CHEN J, YU X, et al. Suppression of methane hydrate dissociation from SDS-dry solution hydrate formation system by a covering liquid method[J]. Fuel, 2020, 277(4): 118-222.
|
| 20 |
SUN Xiaoliang, ZHOU Guanggang, ZHU Jianwei, et al. Molecular dynamics simulation of methane hydrate decomposition in the presence of alcohol additives[J]. Chemphyschem, 2019, 20(19): 2553-2565.
|
| 21 |
YAGASAKI Takuma, MATSUMOTO Masakazu, ANDOH Yoshimichi, et al. Dissociation of methane hydrate in aqueous NaCl solutions[J]. The Journal of Physical Chemistry B, 2014, 118(40): 11797-11804.
|
| 22 |
CHUN Y G, QING Z H, HAO W, et al. Molecular dynamics simulation on the decomposition of type SII hydrogen hydrate and the performance of tetrahydrofuran as a stabiliser[J]. Molecular Simulation, 2010, 36(8): 474-483.
|
| 23 |
陈雪萍, 张鹏, 吴青柏. “自保护”态CO2水合物分解动力及影响因素[J]. 天然气地球科学, 2020, 31(2): 184-193.
|
|
CHEN Xueping, ZHANG Peng, WU Qingbai. Decomposition dynamics and influencing factors of CO2 hydrate in self-preservation state[J]. Natural Gas Geoscience, 2020, 31(2): 184-193.
|
| 24 |
吴强, 靳凯, 张保勇, 等. 初始分解压力对瓦斯水合物分解特性的影响[J]. 黑龙江科技大学学报, 2018, 28(5): 483-487.
|
|
WU Qiang, JIN Kai, ZHANG Baoyong, et al. Effect of dissociation initial pressure on dissociation characteristics of gas hydrate[J]. Journal of Heilongjiang University of Science and Technology, 2018, 28(5): 483-487.
|
| 25 |
刘唯一. 实验研究盐类对甲烷水合物稳定性的影响[D]. 青岛: 中国石油大学(华东), 2018.
|
|
LIU Weiyi. Experimental study on the effect of salt on the stability of methane hydrate[D]. Qingdao: China University of Petroleum (Huadong), 2018.
|
| 26 |
吴强, 王世海, 张保勇, 等. 瓦斯水合物相平衡测定及分解热计算[J]. 中国矿业大学学报, 2017, 46(4): 748-754.
|
|
WU Qiang, WANG Shihai, ZHANG Baoyong, et al. Measurement of phase equilibrium and calculation of dissociation heat of mine gas hydrate[J]. Journal of China University of Mining & Technology, 2017, 46(4): 748-754.
|
 ), 张兆慧1,2, 刘生浩1,2, 焦雯泽1,2, 王立瑾1,2, 滕亚栋1,2, 刘杰1,2
), 张兆慧1,2, 刘生浩1,2, 焦雯泽1,2, 王立瑾1,2, 滕亚栋1,2, 刘杰1,2
 ), ZHANG Zhaohui1,2, LIU Shenghao1,2, JIAO Wenze1,2, WANG Lijin1,2, TENG Yadong1,2, LIU Jie1,2
), ZHANG Zhaohui1,2, LIU Shenghao1,2, JIAO Wenze1,2, WANG Lijin1,2, TENG Yadong1,2, LIU Jie1,2