Chemical Industry and Engineering Progress ›› 2024, Vol. 43 ›› Issue (5): 2409-2419.DOI: 10.16085/j.issn.1000-6613.2023-2004
• Clean and efficient utilization of fossil energy • Previous Articles
Product evolution and reaction mechanism of low-rank coal pyrolysis based on ReaxFF MD simulation
HUANG Zibo1( ), ZHOU Wenjing1(
), ZHOU Wenjing1( ), WEI Jinjia1,2
), WEI Jinjia1,2
- 1.School of Chemical Engineering and Technology, Xi’an Jiaotong University, Xi’an 710049, Shaanxi, China
2.State Key Laboratory of Multiphase Flow in Power Engineering, Xi’an Jiaotong University, Xi’an 710049, Shaanxi, China
-
Received:2023-11-27Revised:2024-01-07Online:2024-06-15Published:2024-05-15 -
Contact:ZHOU Wenjing
基于ReaxFF MD模拟的低阶煤热解产物演化规律及反应机理
- 1.西安交通大学化学工程与技术学院,陕西 西安 710049
2.西安交通大学动力工程多相流国家重点实验室,陕西 西安 710049
-
通讯作者:周文静 -
作者简介:黄淄博(1995—),男,博士研究生,研究方向为煤与生物质热解。E-mail:zbhuang1128@163.com。 -
基金资助:中国华能集团能源安全技术专项(HNKJ20-H87-03)
CLC Number:
Cite this article
HUANG Zibo, ZHOU Wenjing, WEI Jinjia. Product evolution and reaction mechanism of low-rank coal pyrolysis based on ReaxFF MD simulation[J]. Chemical Industry and Engineering Progress, 2024, 43(5): 2409-2419.
黄淄博, 周文静, 魏进家. 基于ReaxFF MD模拟的低阶煤热解产物演化规律及反应机理[J]. 化工进展, 2024, 43(5): 2409-2419.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: https://hgjz.cip.com.cn/EN/10.16085/j.issn.1000-6613.2023-2004
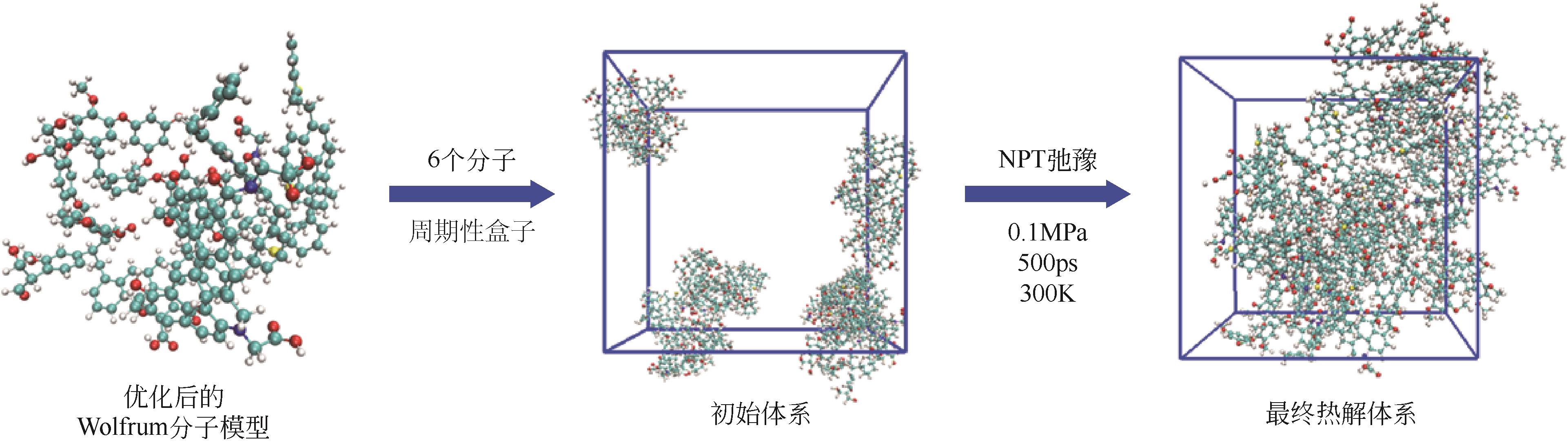
| 状态 | Etotal/kcal·mol–1 | EA/kcal·mol–1 | EB/kcal·mol–1 | ET/kcal·mol–1 | EW/kcal·mol–1 | EH/kcal·mol–1 |
|---|---|---|---|---|---|---|
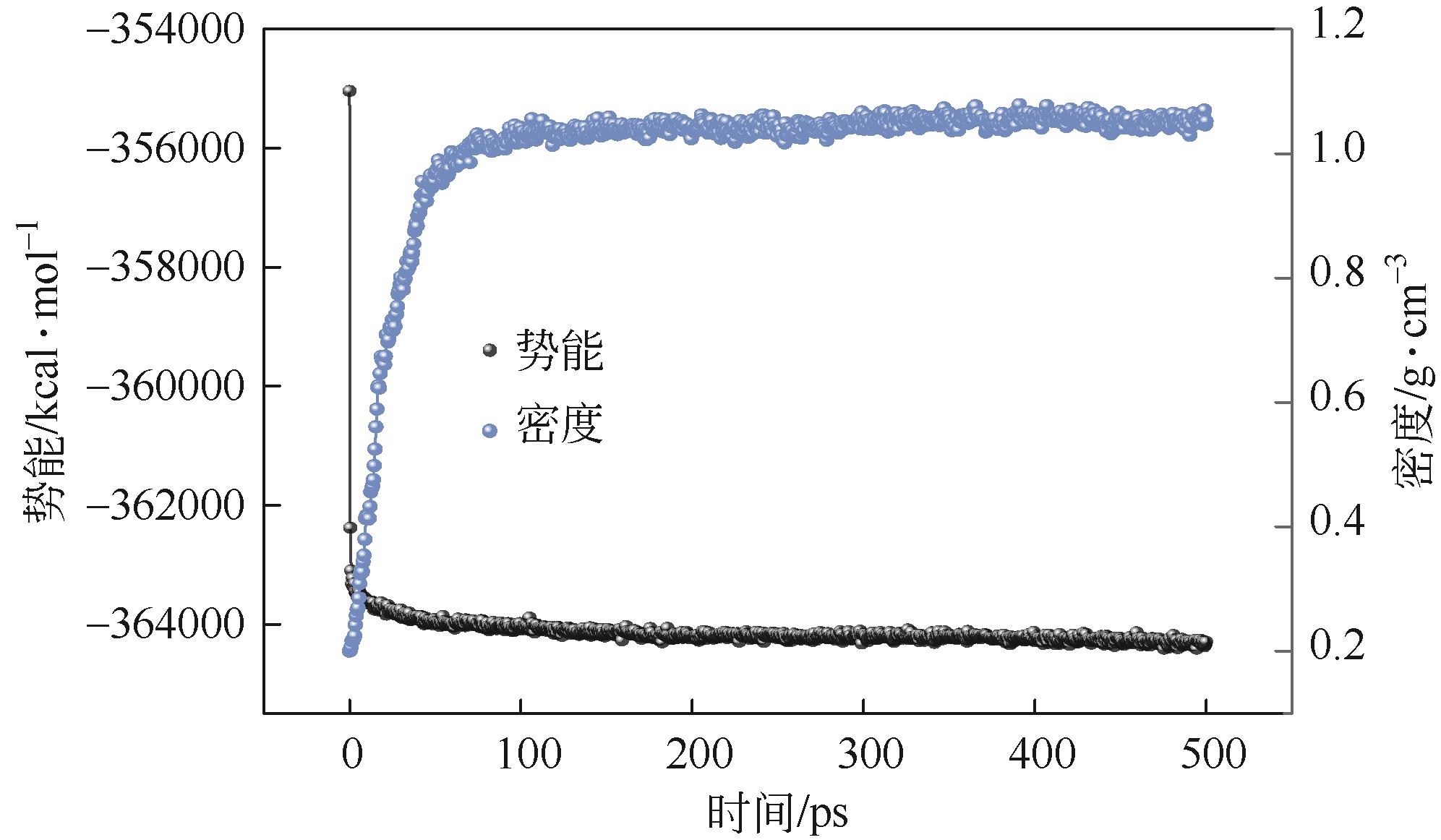
| 初始结构 | 9696.05 | 142.40 | 3719.14 | 279.65 | 5540.87 | –0.30 |
| 几何优化 | 858.17 | 163.41 | 97.53 | 140.86 | 453.71 | –0.84 |
| 退火优化 | 720.90 | 120.75 | 91.97 | 120.75 | 373.64 | –4.68 |
| 状态 | Etotal/kcal·mol–1 | EA/kcal·mol–1 | EB/kcal·mol–1 | ET/kcal·mol–1 | EW/kcal·mol–1 | EH/kcal·mol–1 |
|---|---|---|---|---|---|---|
| 初始结构 | 9696.05 | 142.40 | 3719.14 | 279.65 | 5540.87 | –0.30 |
| 几何优化 | 858.17 | 163.41 | 97.53 | 140.86 | 453.71 | –0.84 |
| 退火优化 | 720.90 | 120.75 | 91.97 | 120.75 | 373.64 | –4.68 |
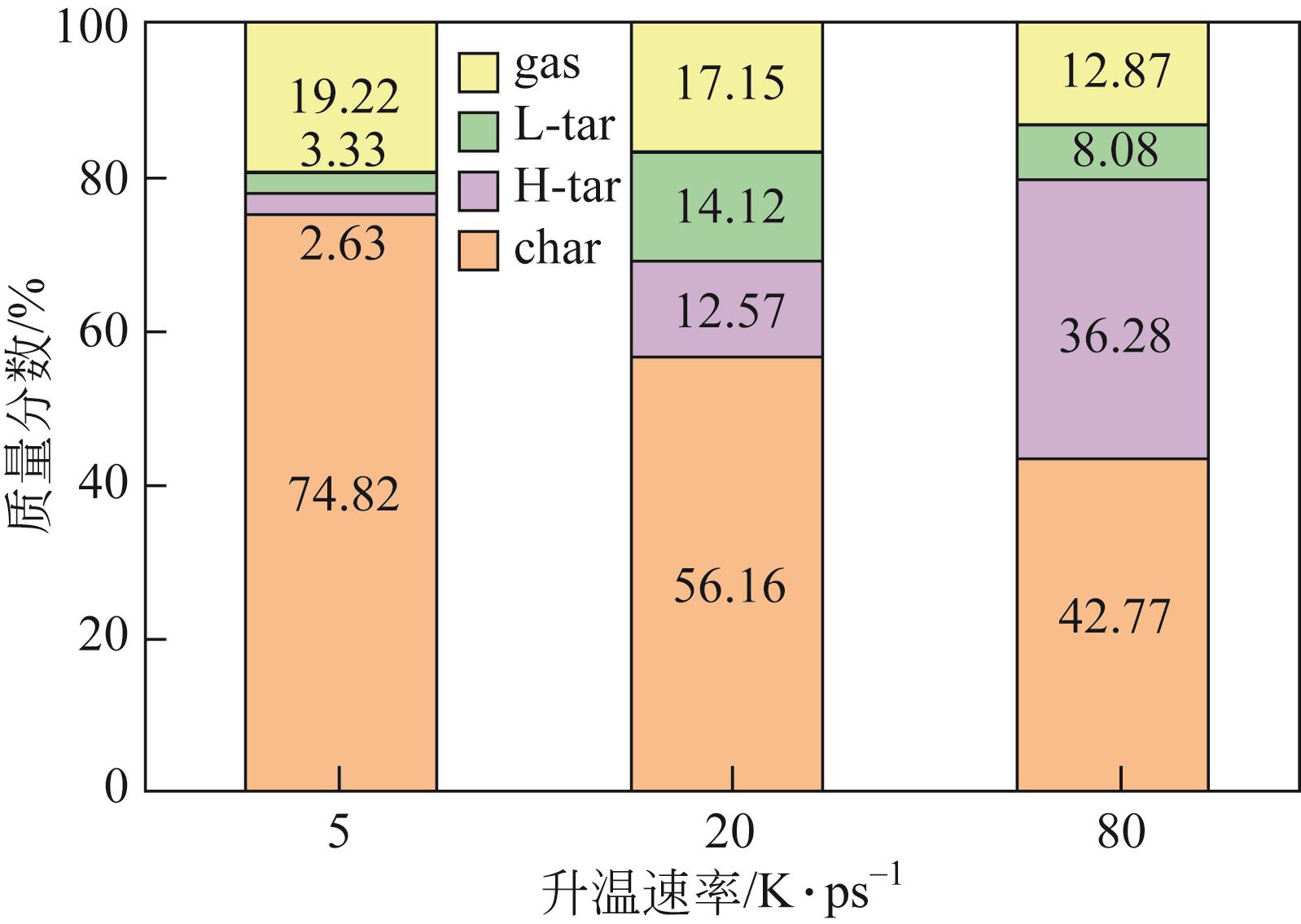
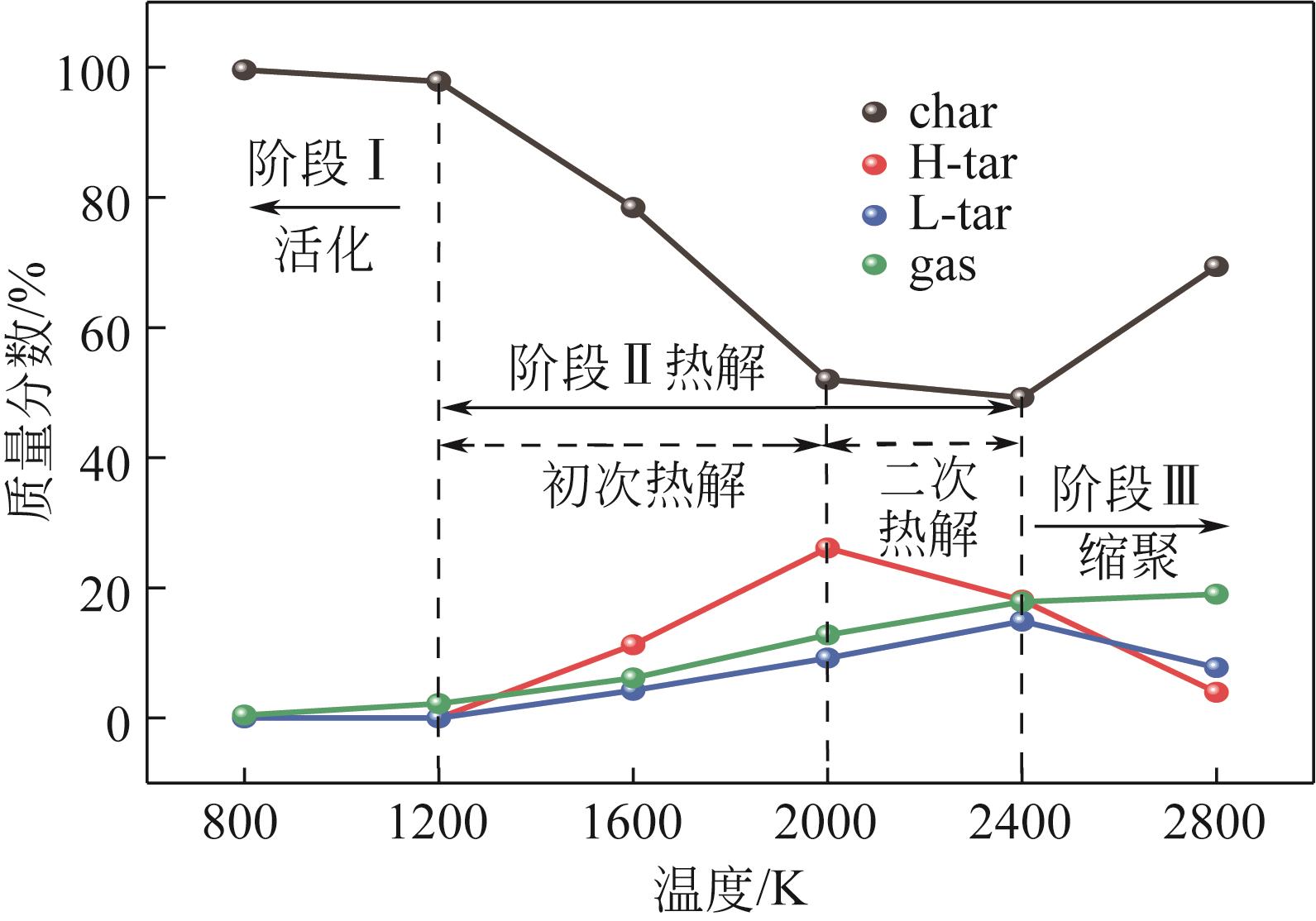
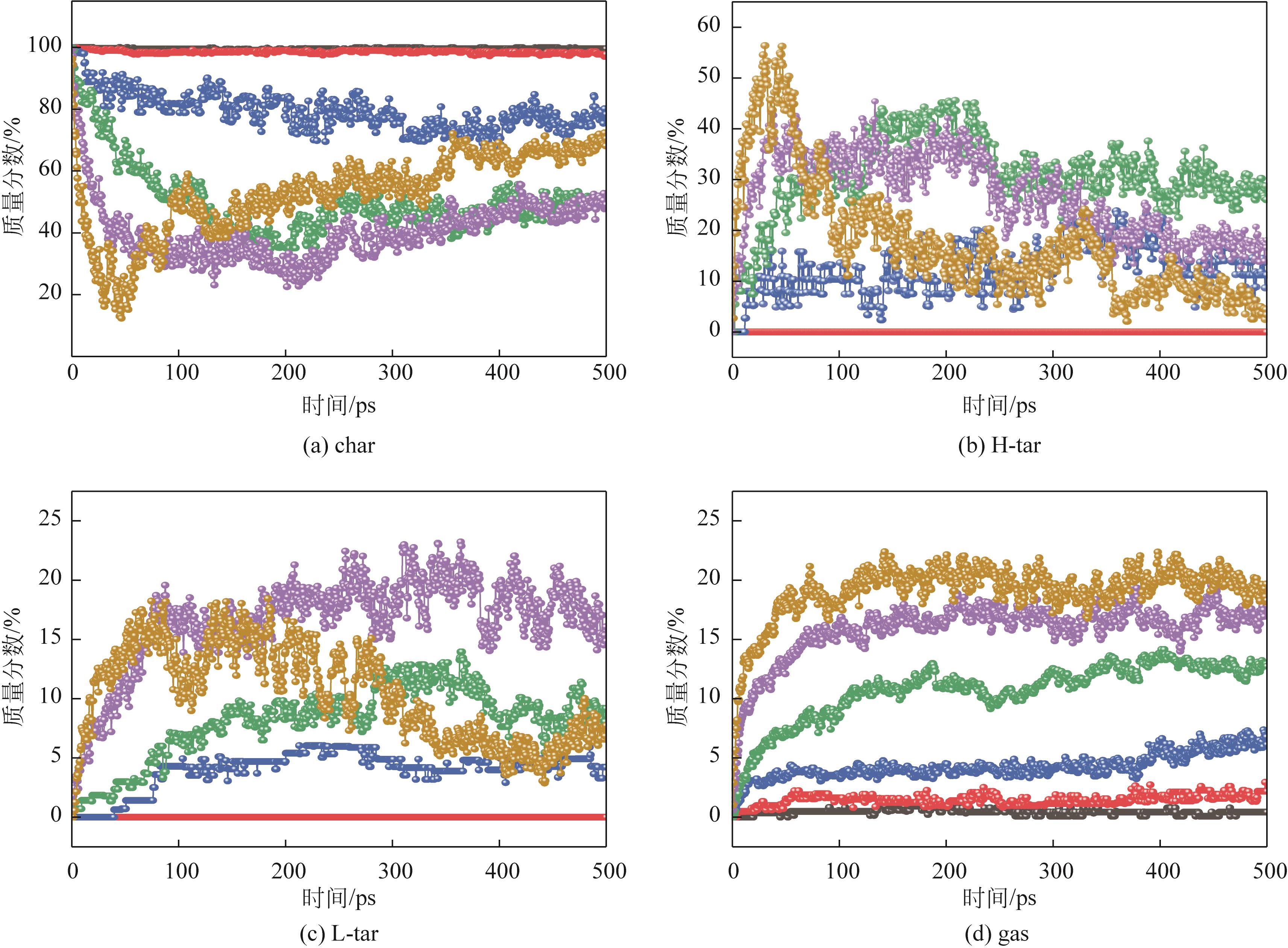
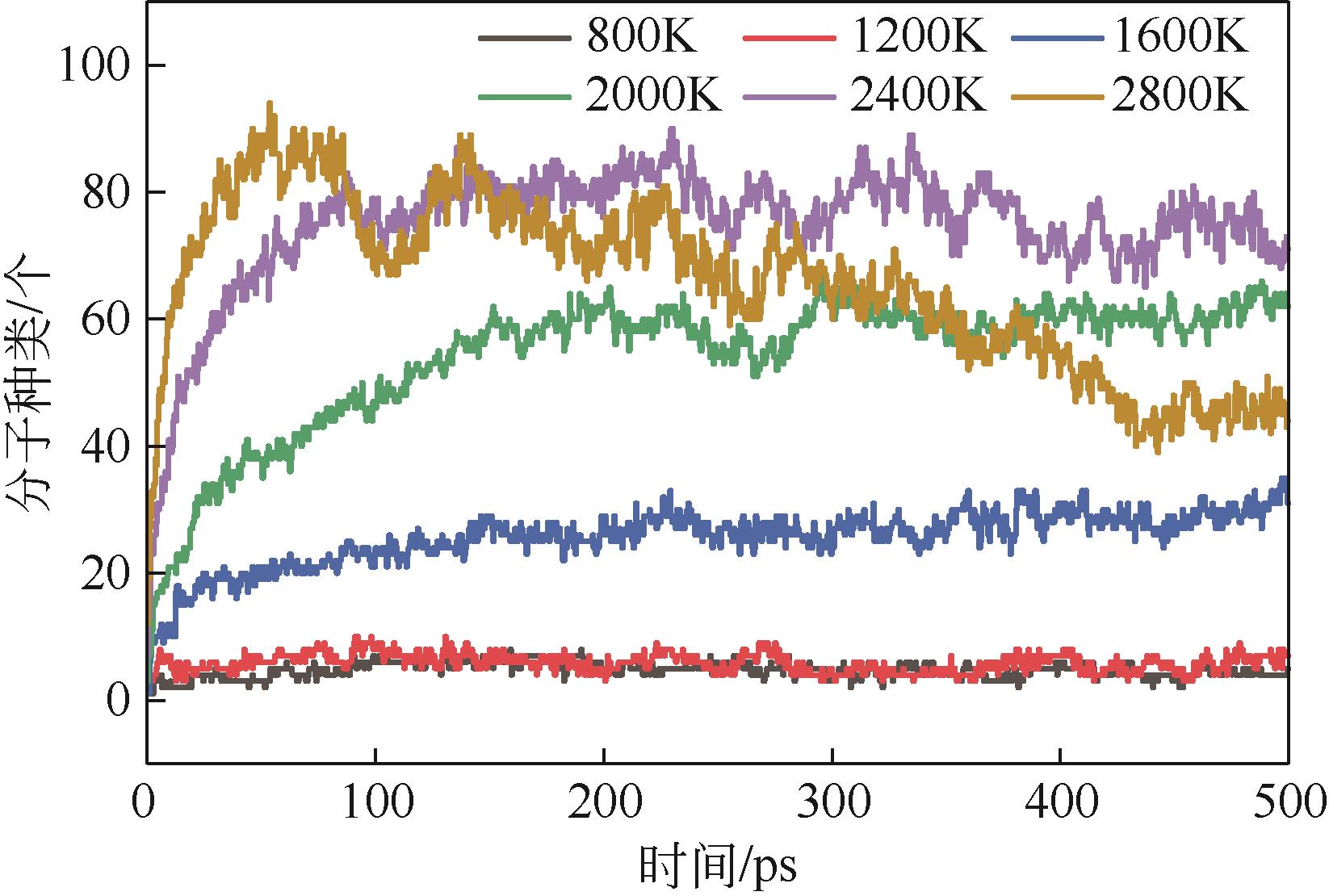
| 物质 | 800K | 1200K | 1600K | 2000K | 2400K | 2800K |
|---|---|---|---|---|---|---|
| char | 2 C225H182O36N4S3 | 1 C225H182O36N4S3 | 1 C217H161O28N3S3 | 1 C135H78O14S2 | 1 C388H211O25N5S2 | 1 C631H307O48N4S4 |
| 2 C225H181O36N4S3 | 1 C223H173O33N4S3 | 1 C187H139O23N3S3 | 1 C113H75O14NS | 1 C131H78O11N2 | 1 C229H103O18S | |
| 2 C225H181O35N4S3 | 1 C222H168O28N4S3 | 1 C186H146O24N3S3 | 1 C72H50O7NS | 1 C60H38O4N | 1 C189H85O13N | |
| 1 C222H177O33N3S3 | 1 C138H96O22S3 | 1 C69H46O4S | 1 C59H37O4 | 1 C48H27O5N | ||
| 1 C221H178O35N2S3 | 1 C137H99O8NS3 | 1 C65H42O8N | 1 C49H27O5S | |||
| 1 C163H142O22N2S3 | 1 C115H76O15N2 | 1 C64H42O10S2 | 1 C45H28O2S | |||
| 1 C66H44O15N2 | 1 C66H52O16N2 | 1 C62H38O6N2S | ||||
| 1 C52H33O2S3 | 1 C60H40O6S2 | |||||
| 1 C59H42O13 | ||||||
| 1 C52H30O4 | ||||||
| H-tar | 1 C39H31ON | 1 C39H25O7N2 | 1 C38H27N | 1 C32H13O4 | ||
| 1 C33H32O7N | 1 C38H29ON | 1 C33H17O3N | 1 C20H16O | |||
| 1 C33H26O4N2 | 1 C38H30ON | 1 C28H19O2N | ||||
| 1 C30H19ON2 | 1 C38H30N | 1 C28H16O | ||||
| 1 C29H19O2N | 1 C38H29O4N | 1 C27H20ON | ||||
| 1 C36H31O2N | 1 C27H12O2N | |||||
| 1 C35H21O3S | 1 C23H13O4 | |||||
| 1 C31H21O3N2 | 1 C20H12O2 | |||||
| 1 C31H20O3N | 1 C17H12O2 | |||||
| 1 C30H19O2N | ||||||
| 1 C17H15S | ||||||
| 1 C17H10O4 | ||||||
| L-tar | 1 C13H13O3 | 1 C13H6O5 | 1 C14H12O2 | 1 C12H5O3S | ||
| 1 C13H14O2 | 1 C11H8O2 | 1 C14H7O7S | 1 C12H5O3 | |||
| 1 C7H7O3 | 1 C11H7O4S | 1 C12H10 | 1 C11H6O2 | |||
| 1 C7H6O3 | 1 C9H8S2 | 1 C12H10O2 | 1 C8H7O | |||
| 1 C6H3O3 | 1 C9H8S | 1 C12H9O5 | 1 C8H4OS | |||
| 1 C5H3O2 | 1 C9H8 | 1 C11H9 | 1 C7H8 | |||
| 1 C4H6O3N | 1 C9H7O | 1 C11H8O | 1 C7H3O5 | |||
| 1 C9H7S | 1 C11H7O | 1 C6H8 | ||||
| 1 C9H7 | 1 C11H6O5S | 1 C5H6 | ||||
| 1 C7H6O | 1 C11H6O2 | |||||
| 1 C7H3O3 | 1 C11H5O4 | |||||
| 1 C6H5O | 1 C10H6O3 | |||||
| 1 C10H5O2 | ||||||
| 1 C9H6O2 | ||||||
| 1 C9H6OS | ||||||
| 2 C9H5O2 | ||||||
| 1 C8H5O2 | ||||||
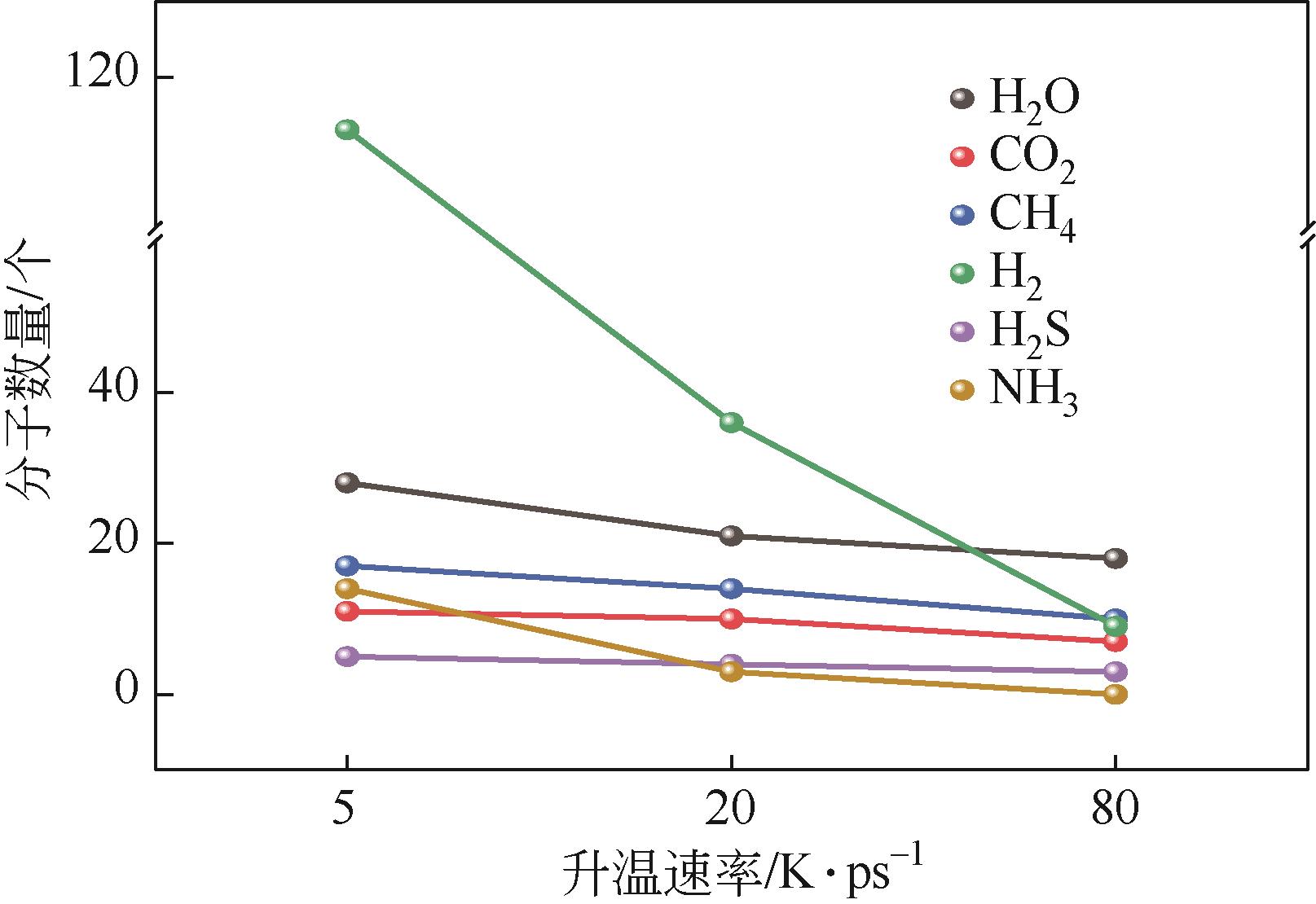
| gas | 2 H2O | 1 C2H4O2N | 1 C3H6O2 | 1 C3H3O | 2 C3H3O | 1 C3HO2 |
| 6 CH2O | 1 C2H5O | 1 C3H4O | 1 C3H3 | 1 C2H4O2 | ||
| 6 H2O | 1 C2H3O2 | 1 C3O4 | 1 C3H2O3 | 2 C2H2O | ||
| 3 C2H3O | 2 C2H5O | 1 C2H3O2 | 1 C2O3 | |||
| 2 C2H2O2 | 1 C2H4O | 2 C2H3O | 1 C2O2S | |||
| 10 CH2O | 3 C2H3O2 | 2 C2H6 | 3 CH2O2 | |||
| 1 CH4O | 1 C2H3OS | 1 C2H4 | 7 CH2O | |||
| 1 CHO | 1 C2H3O | 5 C2H2O | 1 CH3O | |||
| 2 CH4 | 1 C2HO3 | 5 CH2O | 2 CHOS | |||
| 1 CH3 | 3 C2H2O | 10 CHO2 | 16 CHO | |||
| 1 CO2 | 1 CH6N | 1 CH4S | 1 CH3S | |||
| 2 NH3 | 1 CH3O | 2 CHOS | 1 CH2N | |||
| 18 H2O | 4 CH4O | 14 CHO | 19 CH4 | |||
| 3 H2 | 1 CH4N | 18 CH4 | 2 CH3 | |||
| 3 CH3N | 7 CO2 | 1 CH2N | ||||
| 5 CH2O | 1 NH4 | 15 NH3 | ||||
| 2 CHO2 | 9 NH3 | 1 NH2 | ||||
| 11 CHO | 22 H2O | 7 H2S | ||||
| 16 CH4 | 7 H2S | 18 CO2 | ||||
| 1 CH3 | 39 H2 | 1 CO | ||||
| 5 CO2 | 25 H2O | |||||
| 3 NH3 | 108 H2 | |||||
| 20 H2O | 1 H | |||||
| 10 H2 |
| 物质 | 800K | 1200K | 1600K | 2000K | 2400K | 2800K |
|---|---|---|---|---|---|---|
| char | 2 C225H182O36N4S3 | 1 C225H182O36N4S3 | 1 C217H161O28N3S3 | 1 C135H78O14S2 | 1 C388H211O25N5S2 | 1 C631H307O48N4S4 |
| 2 C225H181O36N4S3 | 1 C223H173O33N4S3 | 1 C187H139O23N3S3 | 1 C113H75O14NS | 1 C131H78O11N2 | 1 C229H103O18S | |
| 2 C225H181O35N4S3 | 1 C222H168O28N4S3 | 1 C186H146O24N3S3 | 1 C72H50O7NS | 1 C60H38O4N | 1 C189H85O13N | |
| 1 C222H177O33N3S3 | 1 C138H96O22S3 | 1 C69H46O4S | 1 C59H37O4 | 1 C48H27O5N | ||
| 1 C221H178O35N2S3 | 1 C137H99O8NS3 | 1 C65H42O8N | 1 C49H27O5S | |||
| 1 C163H142O22N2S3 | 1 C115H76O15N2 | 1 C64H42O10S2 | 1 C45H28O2S | |||
| 1 C66H44O15N2 | 1 C66H52O16N2 | 1 C62H38O6N2S | ||||
| 1 C52H33O2S3 | 1 C60H40O6S2 | |||||
| 1 C59H42O13 | ||||||
| 1 C52H30O4 | ||||||
| H-tar | 1 C39H31ON | 1 C39H25O7N2 | 1 C38H27N | 1 C32H13O4 | ||
| 1 C33H32O7N | 1 C38H29ON | 1 C33H17O3N | 1 C20H16O | |||
| 1 C33H26O4N2 | 1 C38H30ON | 1 C28H19O2N | ||||
| 1 C30H19ON2 | 1 C38H30N | 1 C28H16O | ||||
| 1 C29H19O2N | 1 C38H29O4N | 1 C27H20ON | ||||
| 1 C36H31O2N | 1 C27H12O2N | |||||
| 1 C35H21O3S | 1 C23H13O4 | |||||
| 1 C31H21O3N2 | 1 C20H12O2 | |||||
| 1 C31H20O3N | 1 C17H12O2 | |||||
| 1 C30H19O2N | ||||||
| 1 C17H15S | ||||||
| 1 C17H10O4 | ||||||
| L-tar | 1 C13H13O3 | 1 C13H6O5 | 1 C14H12O2 | 1 C12H5O3S | ||
| 1 C13H14O2 | 1 C11H8O2 | 1 C14H7O7S | 1 C12H5O3 | |||
| 1 C7H7O3 | 1 C11H7O4S | 1 C12H10 | 1 C11H6O2 | |||
| 1 C7H6O3 | 1 C9H8S2 | 1 C12H10O2 | 1 C8H7O | |||
| 1 C6H3O3 | 1 C9H8S | 1 C12H9O5 | 1 C8H4OS | |||
| 1 C5H3O2 | 1 C9H8 | 1 C11H9 | 1 C7H8 | |||
| 1 C4H6O3N | 1 C9H7O | 1 C11H8O | 1 C7H3O5 | |||
| 1 C9H7S | 1 C11H7O | 1 C6H8 | ||||
| 1 C9H7 | 1 C11H6O5S | 1 C5H6 | ||||
| 1 C7H6O | 1 C11H6O2 | |||||
| 1 C7H3O3 | 1 C11H5O4 | |||||
| 1 C6H5O | 1 C10H6O3 | |||||
| 1 C10H5O2 | ||||||
| 1 C9H6O2 | ||||||
| 1 C9H6OS | ||||||
| 2 C9H5O2 | ||||||
| 1 C8H5O2 | ||||||
| gas | 2 H2O | 1 C2H4O2N | 1 C3H6O2 | 1 C3H3O | 2 C3H3O | 1 C3HO2 |
| 6 CH2O | 1 C2H5O | 1 C3H4O | 1 C3H3 | 1 C2H4O2 | ||
| 6 H2O | 1 C2H3O2 | 1 C3O4 | 1 C3H2O3 | 2 C2H2O | ||
| 3 C2H3O | 2 C2H5O | 1 C2H3O2 | 1 C2O3 | |||
| 2 C2H2O2 | 1 C2H4O | 2 C2H3O | 1 C2O2S | |||
| 10 CH2O | 3 C2H3O2 | 2 C2H6 | 3 CH2O2 | |||
| 1 CH4O | 1 C2H3OS | 1 C2H4 | 7 CH2O | |||
| 1 CHO | 1 C2H3O | 5 C2H2O | 1 CH3O | |||
| 2 CH4 | 1 C2HO3 | 5 CH2O | 2 CHOS | |||
| 1 CH3 | 3 C2H2O | 10 CHO2 | 16 CHO | |||
| 1 CO2 | 1 CH6N | 1 CH4S | 1 CH3S | |||
| 2 NH3 | 1 CH3O | 2 CHOS | 1 CH2N | |||
| 18 H2O | 4 CH4O | 14 CHO | 19 CH4 | |||
| 3 H2 | 1 CH4N | 18 CH4 | 2 CH3 | |||
| 3 CH3N | 7 CO2 | 1 CH2N | ||||
| 5 CH2O | 1 NH4 | 15 NH3 | ||||
| 2 CHO2 | 9 NH3 | 1 NH2 | ||||
| 11 CHO | 22 H2O | 7 H2S | ||||
| 16 CH4 | 7 H2S | 18 CO2 | ||||
| 1 CH3 | 39 H2 | 1 CO | ||||
| 5 CO2 | 25 H2O | |||||
| 3 NH3 | 108 H2 | |||||
| 20 H2O | 1 H | |||||
| 10 H2 |
| 1 | YU Jiangdong, JIANG Chunyan, GUAN Qingqing, et al. Conversion of low-grade coals in sub- and supercritical water: A review[J]. Fuel, 2018, 217: 275-284. |
| 2 | WANG Chufan, FAN Xing, DONG Xueming, et al. Insights into the structural characteristics of four thermal dissolution extracts of a subbituminous coal by using higher-energy collisional dissociation[J]. Fuel, 2020, 282: 118844. |
| 3 | YANG Fang, DIAN Jie. Macro-economic impact of policies for controlling fossil energy consumption in China[J]. Energies, 2022, 15(3): 1051. |
| 4 | SOLOMON P R, FLETCHER T H, PUGMIRE R J. Progress in coal pyrolysis[J]. Fuel, 1993, 72(5): 587-597. |
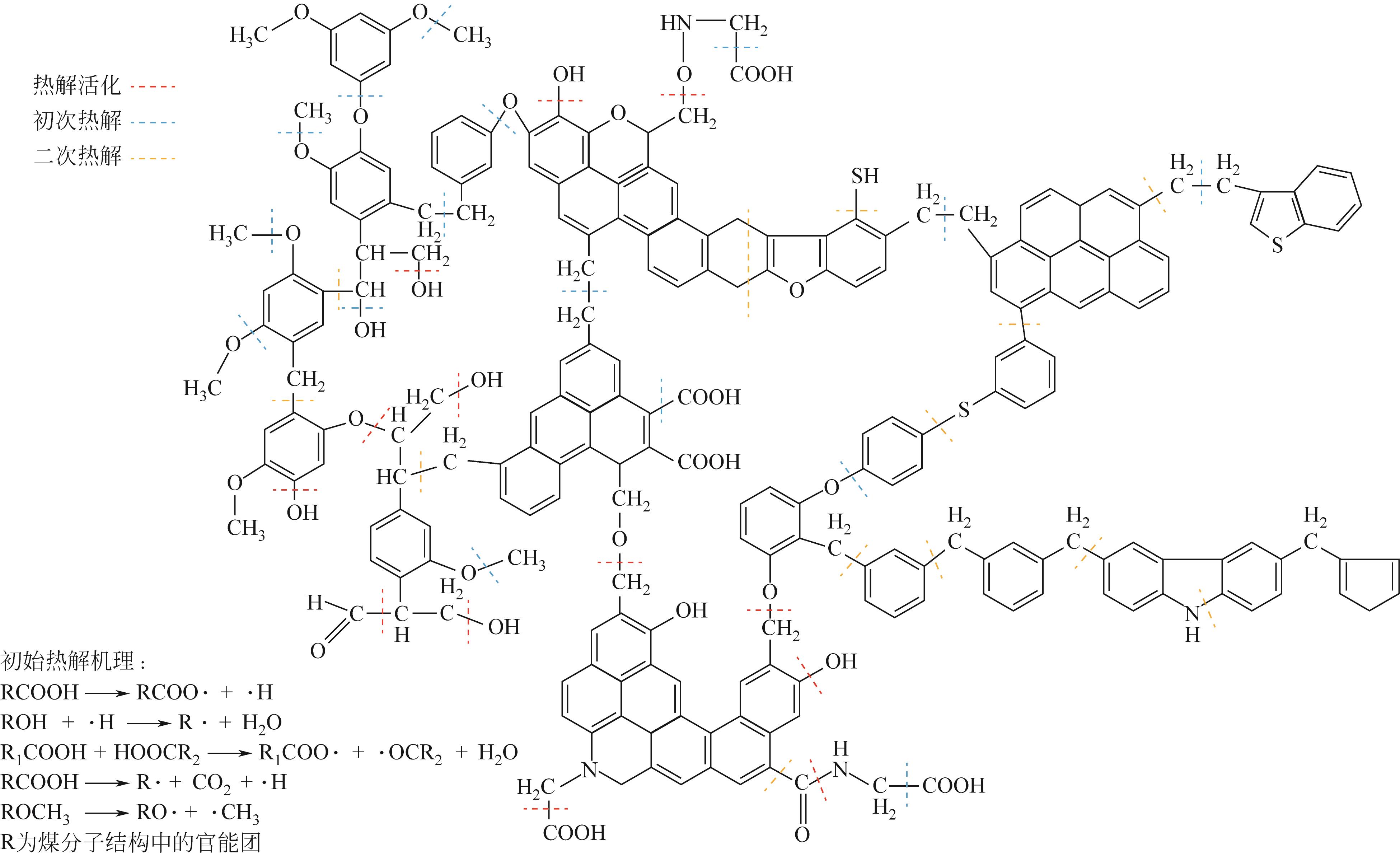
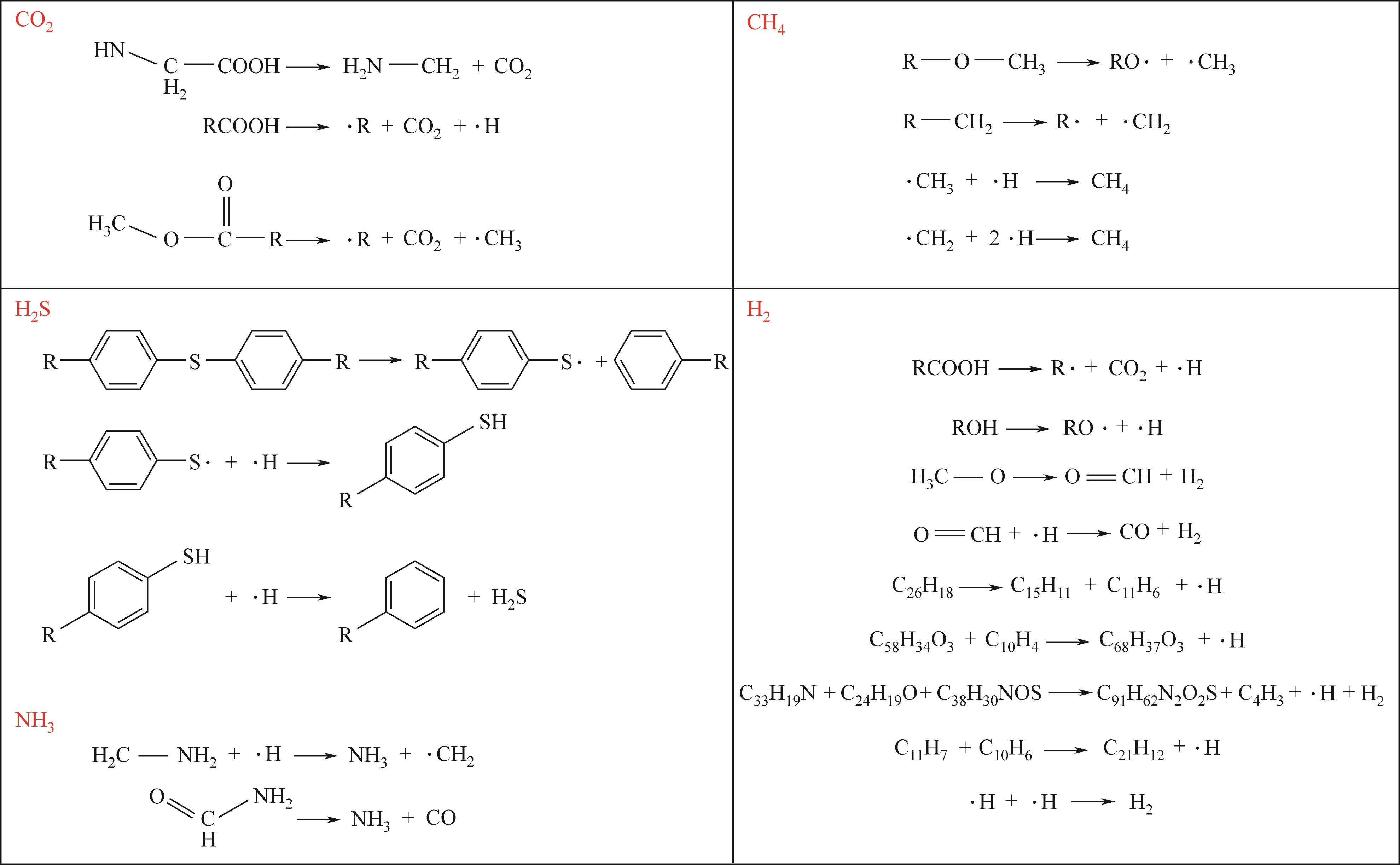
| 5 | GAO Mingjie, LI Xiaoxia, GUO Li. Pyrolysis simulations of Fugu coal by large-scale ReaxFF molecular dynamics[J]. Fuel Processing Technology, 2018, 178: 197-205. |
| 6 | 靳立军, 李扬, 胡浩权. 甲烷活化与煤热解耦合过程提高焦油产率研究进展[J]. 化工学报, 2017, 68(10): 3669-3677. |
| JIN Lijun, LI Yang, HU Haoquan. Research progress of integrated methane activation with coal pyrolysis for improving coal tar yield[J]. CIESC Journal, 2017, 68(10): 3669-3677. | |
| 7 | HONG Dikun, GUO Xin. Molecular dynamics simulations of Zhundong coal pyrolysis using reactive force field[J]. Fuel, 2017, 210: 58-66. |
| 8 | 王芳, 曾玺, 王婷婷, 等. 基于过程强化与反应调控的煤定向热解制高品质油气产物基础研究及中试验证[J]. 化工学报, 2021, 72(12): 6131-6143. |
| WANG Fang, ZENG Xi, WANG Tingting, et al. Fundamentals and pilot demonstration of coal directional pyrolysis to high quality tar and gas products based on process intensification and reaction regulation[J]. CIESC Journal, 2021, 72(12): 6131-6143. | |
| 9 | SOLOMON Peter R, SERIO Michael A, CARANGELO Robert M, et al. Very rapid coal pyrolysis[J]. Fuel, 1986, 65(2): 182-194. |
| 10 | SOLOMON Peter R, SERIO Michael A, SUUBERG Eric M. Coal pyrolysis-experiments, kinetic rates and mechanisms[J]. Progress in Energy and Combustion Science, 1992, 18(2): 133—220. |
| 11 | YU Jianglong, TAHMASEBI Arash, HAN Yanna, et al. A review on water in low rank coals: The existence, interaction with coal structure and effects on coal utilization[J]. Fuel Processing Technology, 2013, 106: 9-20. |
| 12 | 刘方刚, 靳立军, 杨静, 等. 双电离源飞行时间质谱用于峰峰煤原位热解挥发分的表征[J]. 燃料化学学报, 2021, 49(5): 573-581, 564. |
| LIU Fanggang, JIN Lijun, YANG Jing, et al. In-situ characterization of volatiles from pyrolysis of Fengfeng coal by a double ionization time-of-flight mass spectrometer[J]. Journal of Fuel Chemistry and Technology, 2021, 49(5): 573-581, 564. | |
| 13 | FENG Dongdong, SHANG Qi, DONG Heming, et al. Catalytic mechanism of Na on coal pyrolysis-derived carbon black formation: Experiment and DFT simulation[J]. Fuel Processing Technology, 2021, 224: 107011. |
| 14 | VAN DUIN Adri C T, DASGUPTA Siddharth, LORANT Francois, et al. ReaxFF: A reactive force field for hydrocarbons[J]. The Journal of Physical Chemistry A, 2001, 105(41): 9396-9409. |
| 15 | SENFTLE Thomas P, HONG Sungwook, ISLAM Mahbubul Md, et al. The ReaxFF reactive force-field: Development, applications and future directions[J]. npj Computational Materials, 2016, 2(1): 1-14. |
| 16 | 张秀霞, 吕晓雪, 肖美华, 等. 典型烟煤热解机理的反应动力学模拟[J]. 燃料化学学报, 2020, 48(9): 1035-1046. |
| ZHANG Xiuxia, Xiaoxue LYU, XIAO Meihua, et al. Molecular reaction dynamics simulation of pyrolysis mechanism of typical bituminous coal via ReaxFF[J]. Journal of Fuel Chemistry and Technology, 2020, 48(9): 1035-1046. | |
| 17 | CHENOWETH Kimberly, VAN DUIN Adri C T, GODDARD William A. ReaxFF reactive force field for molecular dynamics simulations of hydrocarbon oxidation[J]. The Journal of Physical Chemistry A, 2008, 112(5): 1040-1053. |
| 18 | SALMON Elodie, VAN DUIN Adri C T, LORANT François, et al. Early maturation processes in coal. Part 2: Reactive dynamics simulations using the ReaxFF reactive force field on Morwell Brown coal structures[J]. Organic Geochemistry, 2009, 40(12): 1195-1209. |
| 19 | BHOI Sanjukta, BANERJEE Tamal, MOHANTY Kaustubha. Molecular dynamic simulation of spontaneous combustion and pyrolysis of brown coal using ReaxFF[J]. Fuel, 2014, 136: 326-333. |
| 20 | Fidel CASTRO-MARCANO, KAMAT Amar M, RUSSO Michael F, et al. Combustion of an Illinois No. 6 coal char simulated using an atomistic char representation and the ReaxFF reactive force field[J]. Combustion and Flame, 2012, 159(3): 1272-1285. |
| 21 | 冯炜, 高红凤, 王贵, 等. 枣泉煤分子模型构建及热解的分子模拟[J]. 化工学报, 2019, 70(4): 1522-1531. |
| FENG Wei, GAO Hongfeng, WANG Gui, et al. Molecular model and pyrolysis simulation of Zaoquan coal[J]. CIESC Journal, 2019, 70(4): 1522-1531. | |
| 22 | 郑默, 李晓霞. ReaxFF MD模拟揭示的煤热解挥发分自由基反应的竞争与协调[J]. 化工学报, 2022, 73(6): 2732-2741. |
| ZHENG Mo, LI Xiaoxia. Revealing reaction compromise in competition for volatile radicals during coal pryolysis via ReaxFF MD simulation[J]. CIESC Journal, 2022, 73(6): 2732-2741. | |
| 23 | ZHAN Jinhui, WU Rongcheng, LIU Xiaoxing, et al. Preliminary understanding of initial reaction process for subbituminous coal pyrolysis with molecular dynamics simulation[J]. Fuel, 2014, 134: 283-292. |
| 24 | ZHENG Mo, LI Xiaoxia, LIU Jian, et al. Initial chemical reaction simulation of coal pyrolysis via ReaxFF molecular dynamics[J]. Energy & Fuels, 2013, 27(6): 2942-2951. |
| 25 | ZHOU Zhijun, GUO Longzhen, CHEN Liping, et al. Study of pyrolysis of brown coal and gasification of coal-water slurry using the ReaxFF reactive force field[J]. International Journal of Energy Research, 2018, 42(7): 2465-2480. |
| 26 | LIU Lianchi, Andres JARAMILLO-BOTERO, GODDARD William A Ⅲ, et al. Development of a ReaxFF reactive force field for ettringite and study of its mechanical failure modes from reactive dynamics simulations[J]. The Journal of Physical Chemistry A, 2012, 116(15): 3918-3925. |
| 27 | ZHENG Mo, PAN Yang, WANG Ze, et al. Capturing the dynamic profiles of products in Hailaer brown coal pyrolysis with reactive molecular simulations and experiments[J]. Fuel, 2020, 268: 117290. |
| 28 | WANG Jin, HOU Quanlin, ZENG Fangui, et al. Gas generation mechanisms of bituminous coal under shear stress based on ReaxFF molecular dynamics simulation[J]. Fuel, 2021, 298: 120240. |
| 29 | MATHEWS Jonathan P, CHAFFEE Alan L. The molecular representations of coal—A review[J]. Fuel, 2012, 96: 1-14. |
| 30 | JURKIEWICZ Antoni. Spatial system of the Wiser model of coal structure according to the second moment of the nuclear magnetic resonance line[J]. Journal of applied physics, 1987, 62(9): 3892-3897. |
| 31 | SHINN John H. From coal to single-stage and two-stage products: A reactive model of coal structure[J]. Fuel, 1984, 63(9): 1187-1196. |
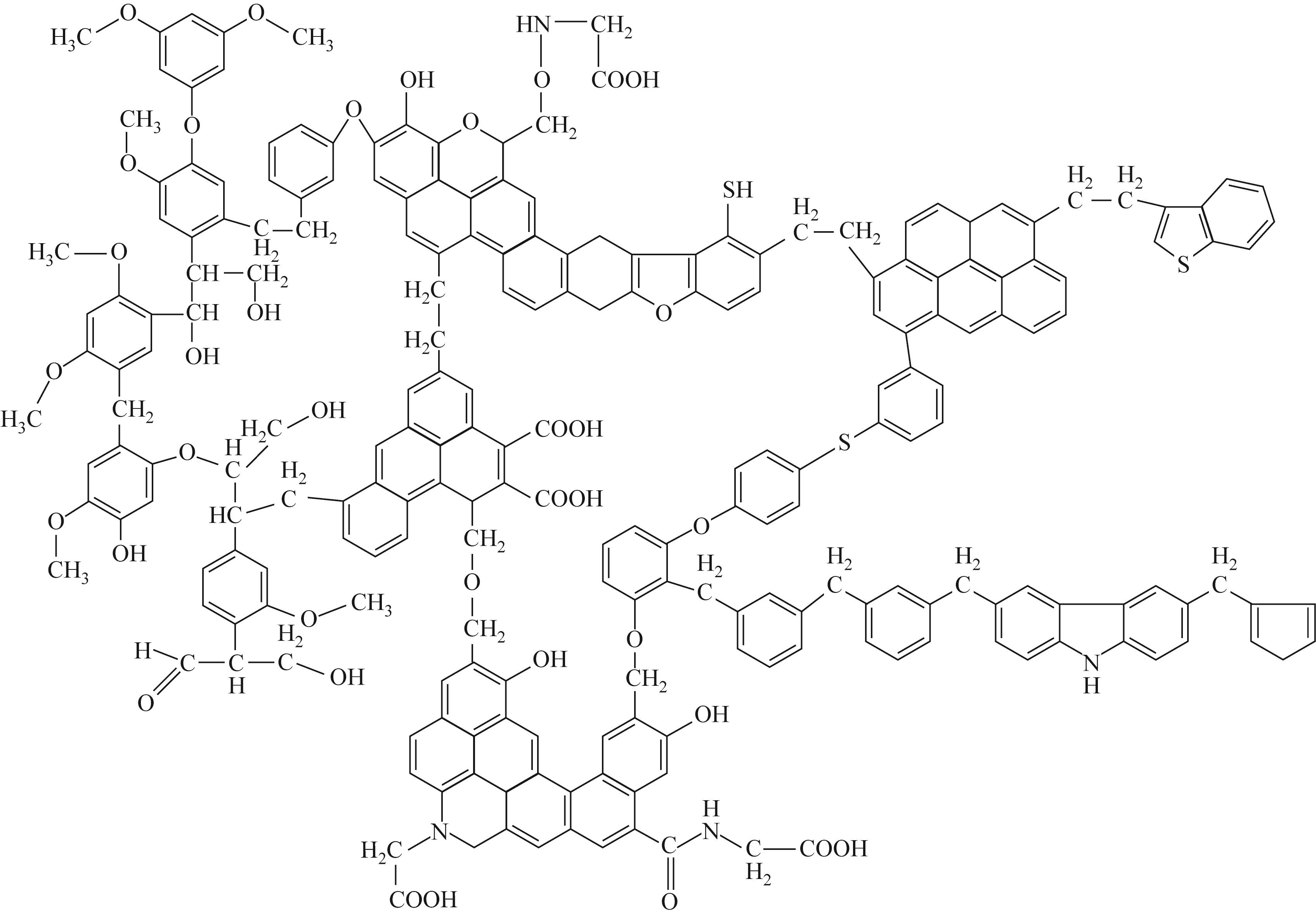
| 32 | WOLFRUM E A. Correlations between petrographical properties, chemical structure, and technological behavior of Rhenish brown coal[M]. Washington, D.C.: American Chemical Society, 1984: 15-37. |
| 33 | LIANG Yinghua, WANG Feng, ZHANG Hang, et al. A Reax FF molecular dynamics study on the mechanism of organic sulfur transformation in the hydropyrolysis process of lignite[J]. Fuel Processing Technology, 2016, 147: 32-40. |
| 34 | ZHENG Mo, LI Xiaoxia, WANG Meijun, et al. Dynamic profiles of tar products during Naomaohu coal pyrolysis revealed by large-scale reactive molecular dynamic simulation[J]. Fuel, 2019, 253: 910-920. |
| 35 | MUELLER Jonathan E, VAN DUIN Adri C T, GODDARD William A Ⅲ. Application of the ReaxFF reactive force field to reactive dynamics of hydrocarbon chemisorption and decomposition[J]. The Journal of Physical Chemistry C, 2010, 114(12): 5675-5685. |
| 36 | Malte DÖNTGEN, Marie-Dominique PRZYBYLSKI-FREUND, KRÖGER Leif C, et al. Automated discovery of reaction pathways, rate constants, and transition states using reactive molecular dynamics simulations[J]. Journal of Chemical Theory and Computation, 2015, 11(6): 2517-2524. |
| 37 | FLETCHER Thomas H, KERSTEIN Alan R, PUGMIRE Ronald J, et al. Chemical percolation model for devolatilization. 3. Direct use of carbon-13 NMR data to predict effects of coal type[J]. Energy & Fuels, 1992, 6(4): 414-431. |
| 38 | SATHE Chirag, PANG Yanyuan, LI Chunzhu. Effects of heating rate and ion-exchangeable cations on the pyrolysis yields from a Victorian brown coal[J]. Energy & Fuels, 1999, 13(3): 748-755. |
| 39 | HAYASHI Jun-ichiro, TAKAHASHI Hiroshi, Satoshi DOI, et al. Reactions in brown coal pyrolysis responsible for heating rate effect on tar yield[J]. Energy & Fuels, 2000, 14(2): 400-408. |
| 40 | LEI Zhao, LIANG Qijun, LING Qiang, et al. Investigating the reaction mechanism of light tar for Shenfu bituminous coal pyrolysis[J]. Energy, 2023, 263: 125731. |
| [1] | WU Qi, BAI Boyang, YIN Yongjie, MA Xiaoxun. Relationship between the structure of macerals of Ordos lignite and its pyrolysis characteristics [J]. Chemical Industry and Engineering Progress, 2024, 43(5): 2370-2385. |
| [2] |
ZHANG Pengfei, YAN Zhangyan, REN Liang, ZHAGN Kui, LIANG Jialin, ZHAO Guangle, ZHANG Fanbin, HU Zhihai.
Research progress in the catalytic hydrodealkylation of C |
| [3] | ZHANG Xin, TANG Jiyun, CHEN Juan, SONG Zhanlong, DONG Yong, YAO Hong. Transformation of trace metals Cu and Pb during high temperature flue gas pyrolysis of waste tires [J]. Chemical Industry and Engineering Progress, 2024, 43(3): 1606-1613. |
| [4] | CHEN Guohui, WANG Junlei, LI Shilong, LI Jinyu, XU Yunfei, LUO Junxiao, WANG Kun. Progress in synthesis of ternary cathode materials for lithium-ion batteries by flame spray pyrolysis [J]. Chemical Industry and Engineering Progress, 2024, 43(2): 971-983. |
| [5] | REN Pengkun, ZHONG Zhaoping, YANG Yuxuan, ZHANG Shan, DU Haoran, LI Qian. Control of heavy metals in sludge pyrolysis process by modified sepiolite [J]. Chemical Industry and Engineering Progress, 2024, 43(1): 541-550. |
| [6] | CHENG Haolin, NIAN Yao, HAN You. Progress in the mechanism of CH4 and CO2co-conversion reactions [J]. Chemical Industry and Engineering Progress, 2024, 43(1): 60-75. |
| [7] | LI Zhiyuan, HUANG Yaji, ZHAO Jiaqi, YU Mengzhu, ZHU Zhicheng, CHENG Haoqiang, SHI Hao, WANG Sheng. Characterization of heavy metals during co-pyrolysis of sludge with PVC [J]. Chemical Industry and Engineering Progress, 2023, 42(9): 4947-4956. |
| [8] | LI Haidong, YANG Yuankun, GUO Shushu, WANG Benjin, YUE Tingting, FU Kaibin, WANG Zhe, HE Shouqin, YAO Jun, CHEN Shu. Effect of carbonization and calcination temperature on As(Ⅲ) removal performance of plant-based Fe-C microelectrolytic materials [J]. Chemical Industry and Engineering Progress, 2023, 42(7): 3652-3663. |
| [9] | YAO Liming, WANG Yazhuo, FAN Honggang, GU Qing, YUAN Haoran, CHEN Yong. Treatment status of kitchen waste and its research progress of pyrolysis technology [J]. Chemical Industry and Engineering Progress, 2023, 42(7): 3791-3801. |
| [10] | ZHANG Shan, ZHONG Zhaoping, YANG Yuxuan, DU Haoran, LI Qian. Enrichment of heavy metals in pyrolysis of municipal solid waste by phosphate modified kaolin [J]. Chemical Industry and Engineering Progress, 2023, 42(7): 3893-3903. |
| [11] | LI Ruolin, HE Shaolin, YUAN Hongying, LIU Boyue, JI Dongli, SONG Yang, LIU Bo, YU Jiqing, XU Yingjun. Effect of in-situ pyrolysis on physical properties of oil shale and groundwater quality [J]. Chemical Industry and Engineering Progress, 2023, 42(6): 3309-3318. |
| [12] | LI Dongxian, WANG Jia, JIANG Jianchun. Producing biofuels from soapstock via pyrolysis and subsequent catalytic vapor-phase hydrotreating process [J]. Chemical Industry and Engineering Progress, 2023, 42(6): 2874-2883. |
| [13] | WANG Zhiwei, GUO Shuaihua, WU Mengge, CHEN Yan, ZHAO Junting, LI Hui, LEI Tingzhou. Recent advances on catalytic co-pyrolysis of biomass and plastic [J]. Chemical Industry and Engineering Progress, 2023, 42(5): 2655-2665. |
| [14] | LIANG Yijing, MA Yan, LU Zhanfeng, QIN Fusheng, WAN Junjie, WANG Zhiyuan. Experimental investigation on the anti-coking performance of La1-x Sr x MnO3 perovskite coating [J]. Chemical Industry and Engineering Progress, 2023, 42(4): 1769-1778. |
| [15] | LIU Jing, LIN Lin, ZHANG Jian, ZHAO Feng. Research progress in pore size regulation and electrochemical performance of biomass-based carbon materials [J]. Chemical Industry and Engineering Progress, 2023, 42(4): 1907-1916. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||