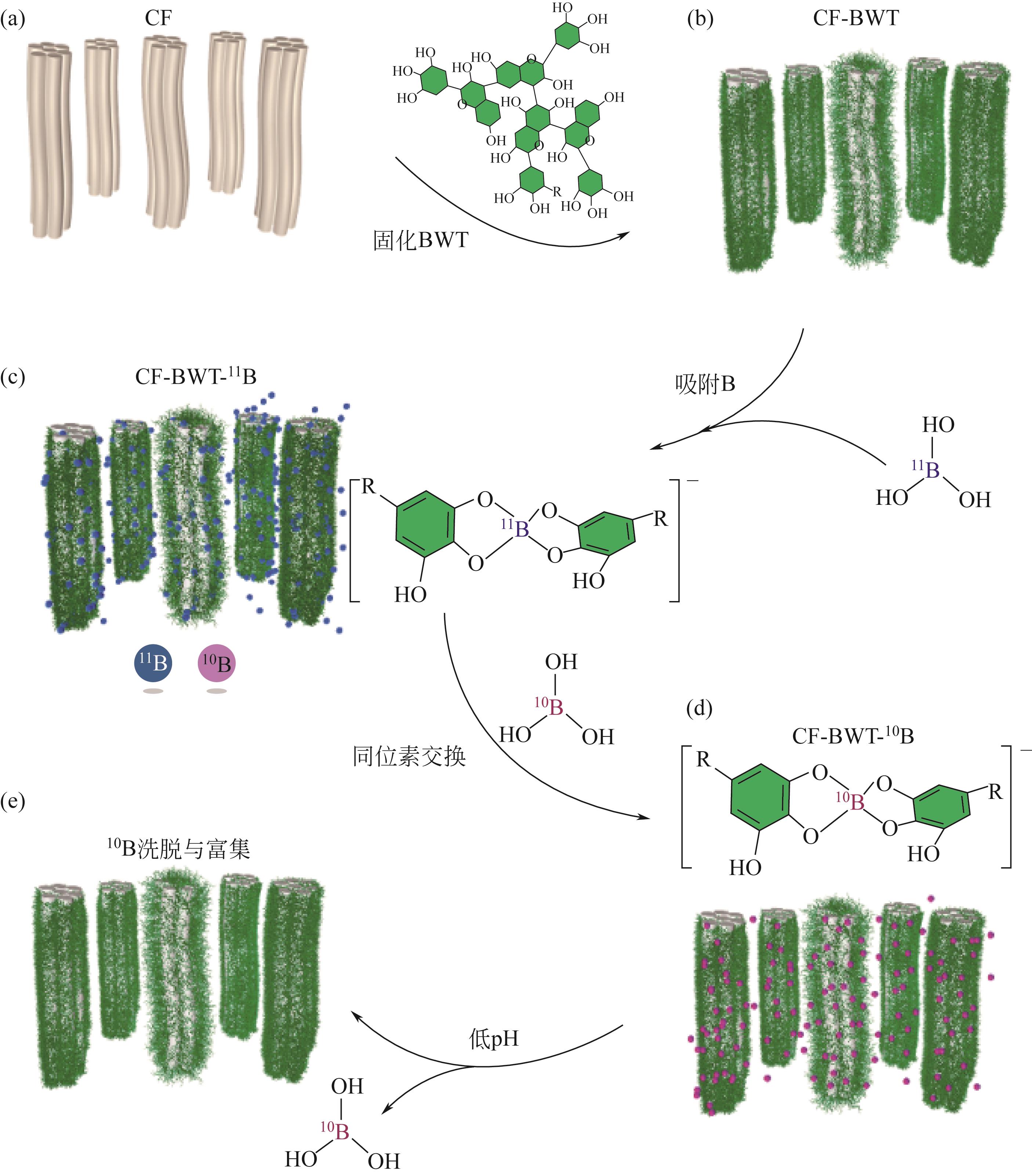
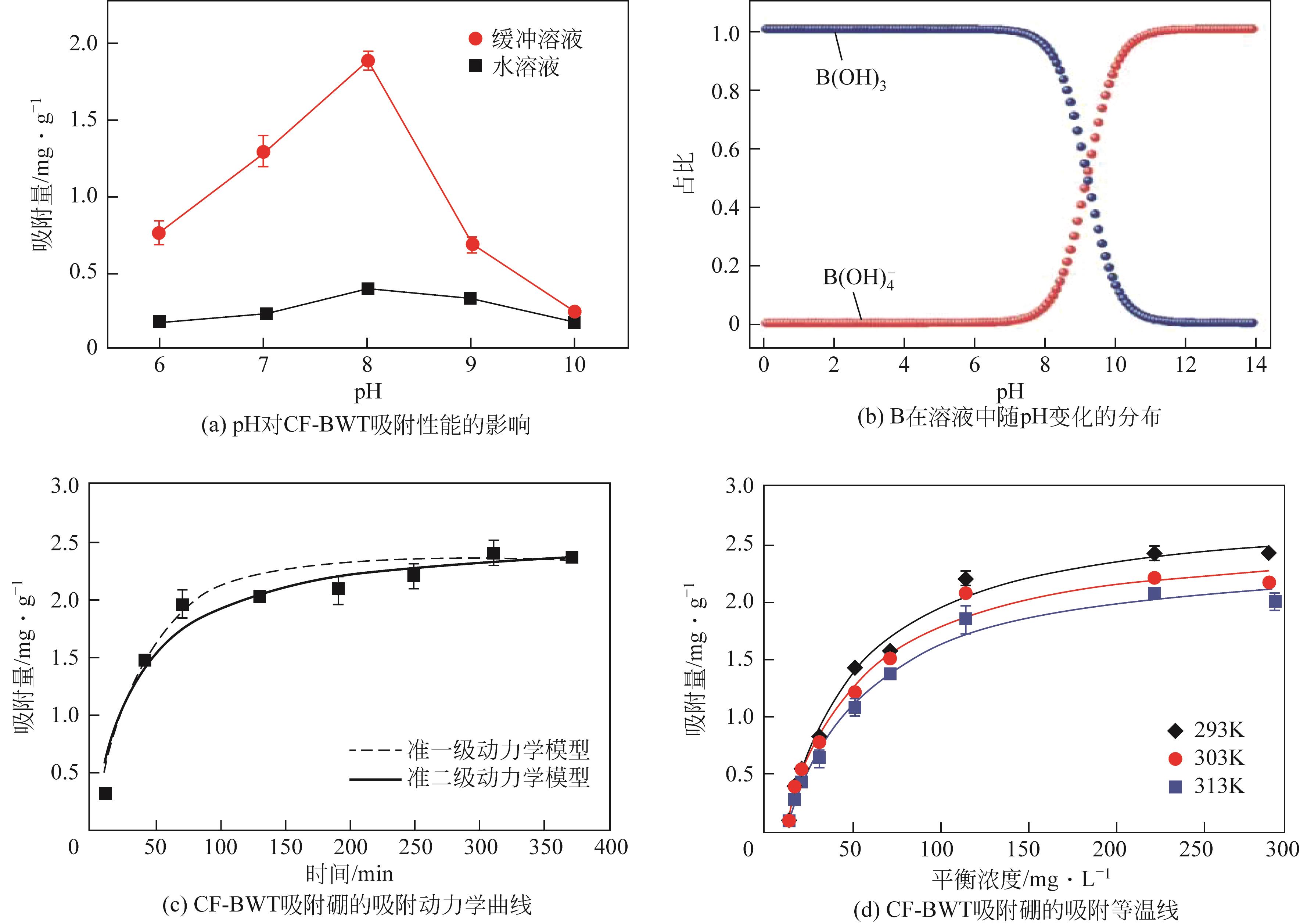
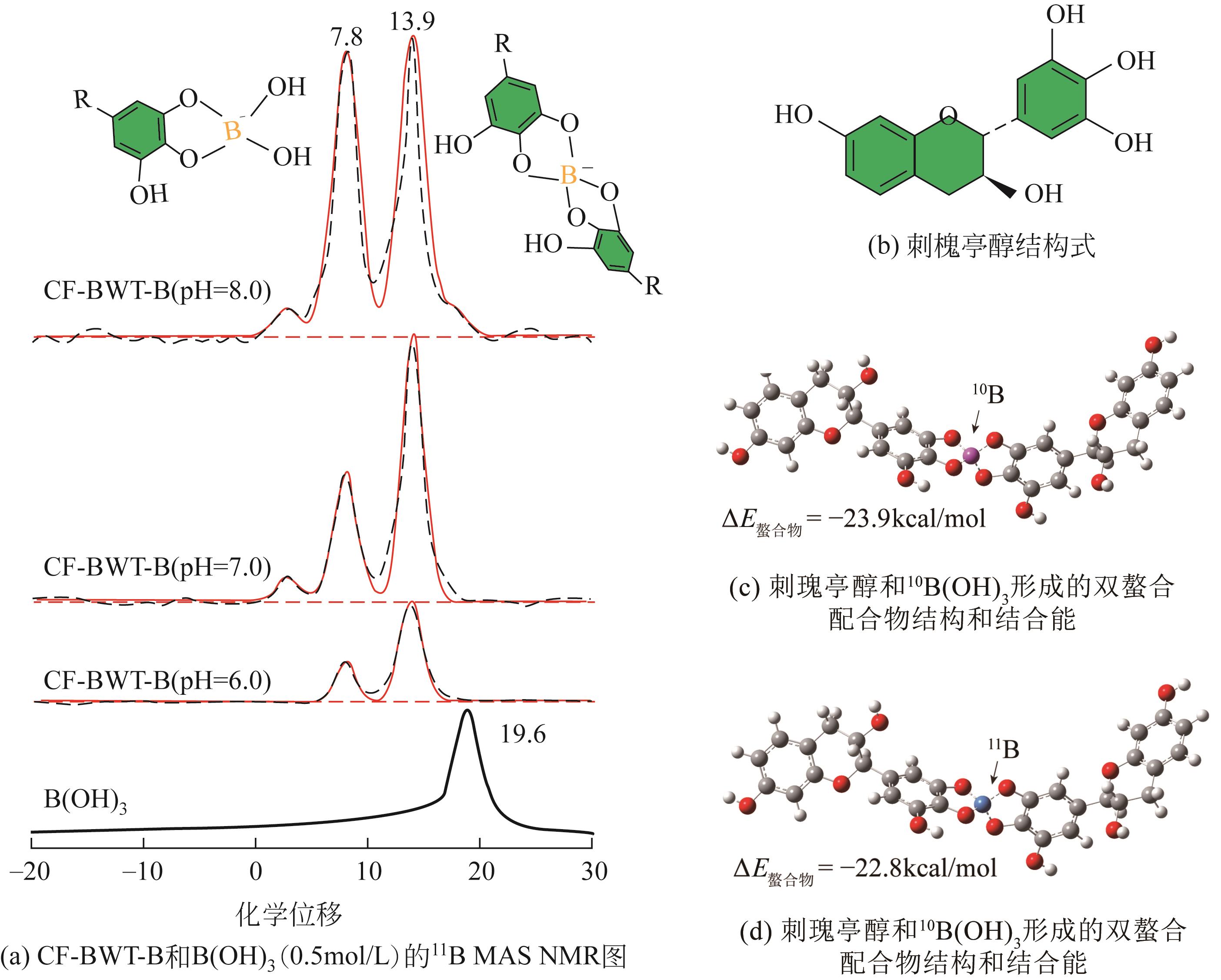
| 1 |
王美玲, 付道贵, 王贯春, 等. 同位素分离理论在硼同位素分离中的应用[J]. 化学工程, 2013, 41: 20-22, 73.
|
|
WANG Meiling, FU Daogui, WANG Guanchun, et al. Application of isotopic separation theory in separation of boron isotopes[J]. Chemical Engineering (China), 2013, 41: 20-22, 73.
|
| 2 |
CHEN Tao, WANG Qingfeng, Jiafei LYU, et al. Boron removal and reclamation by magnetic magnetite (Fe3O4) nanoparticle: An adsorption and isotopic separation study[J]. Separation and Purification Technology, 2020, 231: 115930.
|
| 3 |
IVANOV Vladimir A, KATALNIKOV Sergei G. Physico-chemical and engineering principles of boron isotopes separation by using BF3-anisole·BF3 system[J]. Separation Science and Technology, 2001, 36(8/9): 1737-1768.
|
| 4 |
KAKIHANA Hidetake, KOTAKA Masahiro, SATOH Shohei, et al. Fundamental studies on the ion-exchange separation of boron isotopes[J]. Bulletin of the Chemical Society of Japan, 1977, 50(1): 158-163.
|
| 5 |
LYAKHOV K A, LEE H J, PECHEN A N. Some issues of industrial scale boron isotopes separation by the laser assisted retarded condensation (SILARC) method[J]. Separation and Purification Technology, 2017, 176: 402-411.
|
| 6 |
IVANOV P I, KHOROSHILOV A V, PYANIN D V. Inversion of boron isotope effect during chemical exchange in extraction systems[J]. Journal of Physics: Conference Series, 2020, 1696(1): 012017.
|
| 7 |
Jiafei LYU, ZENG Zhouliangzi, ZHANG Nan, et al. Pyrocatechol-modified resins for boron recovery from water: Synthesis, adsorption, and isotopic separation studies[J]. Reactive and Functional Polymers, 2017, 112: 1-8.
|
| 8 |
AIDA Masao, FUJII Yasuhiko, OKAMOTO Makoto. Chromatographic enrichment of 10B by using weak-base anion-exchange resin[J]. Separation Science and Technology, 1986, 21(6/7): 643-654.
|
| 9 |
WANG Qingfeng, CHEN Tao, BAI Peng, et al. Fe3O4-loaded ion exchange resin for chromatographic separation of boron isotopes: Experiment and numerical simulation[J]. Chemical Engineering Research and Design, 2021, 171: 358-366.
|
| 10 |
Jiafei LYU, LIU Hongxu, ZHANG Jingshuang, et al. Metal-organic frameworks(MOFs) as highly efficient agents for boron removal and boron isotope separation[J]. RSC Advances, 2017, 7: 16022-16026.
|
| 11 |
DZAH Courage Sedem, DUAN Yuqing, ZHANG Haihui, et al. Latest developments in polyphenol recovery and purification from plant by-products: A review[J]. Trends in Food Science & Technology, 2020, 99: 375-388.
|
| 12 |
ZHOU Jiajing, LIN Zhixing, JU Yi, et al. Polyphenol-mediated assembly for particle engineering[J]. Accounts of Chemical Research, 2020, 53(7): 1269-1278.
|
| 13 |
GENG Huimin, ZHONG Qizhi, LI Jianhua, et al. Metal ion-directed functional metal-phenolic materials[J]. Chemical Reviews, 2022, 122(13): 11432-11473.
|
| 14 |
GUO Junling, SUN Huanli, Karen ALT, et al. Boronate-phenolic network capsules with dual response to acidic pH and cis-diols[J]. Advanced Healthcare Materials, 2015, 4(12): 1796-1801.
|
| 15 |
MORISADA Shintaro, Tetsuzen RIN, OGATA Takeshi, et al. Adsorption removal of boron in aqueous solutions by amine-modified tannin gel[J]. Water Research, 2011, 45(13): 4028-4034.
|
| 16 |
SUN Haodong, XIA Nannan, WU Cheng, et al. Efficient and reversible removal of boric acid by chitosan/tannic acid functional paper[J]. BioResources, 2019, 14(4): 7734-7750.
|
| 17 |
CHEN Yuanzhi, Jiafei LYU, WANG Yuming, et al. Synthesis, characterization, adsorption, and isotopic separation studies of pyrocatechol-modified MCM-41 for efficient boron removal[J]. Industrial & Engineering Chemistry Research, 2019, 58(8): 3282-3292.
|
| 18 |
石碧, 狄莹. 植物多酚[M]. 北京: 科学出版社, 2000.
|
|
SHI Bi, DI Ying. Plant polyphenols[M]. Beijing: Science Press, 2000.
|
| 19 |
AUAD Priscila, SPIER Franciela, GUTTERRES Mariliz. Vegetable tannin composition and its association with the leather tanning effect[J]. Chemical Engineering Communications, 2020, 207(5): 722-732.
|
| 20 |
LI Shuangyang, WANG Yaping, XU Weixing, et al. Natural rubber-based elastomer reinforced by chemically modified multiscale leather collagen fibers with excellent toughness[J]. ACS Sustainable Chemistry & Engineering, 2020, 8(13): 5091-5099.
|
| 21 |
Yu MATATOV-MEYTAL, SHEINTUCH M. Catalytic fibers and cloths[J]. Applied Catalysis A: General, 2002, 231(1/2): 1-16.
|
| 22 |
LIAO Xuepin, ZHANG Mina, SHI Bi. Collagen-fiber-immobilized tannins and their adsorption of Au( Ⅲ )[J]. Industrial Engineering Chemistry Research, 2004, 43(9): 2222-2227.
|
| 23 |
MA Jianzhong, LIU Qiwu, WU Mengxin, et al. Preparation and assistant-dyeing of formaldehyde-free amphoteric acrylic retaining agent[J]. Journal of Leather Science and Engineering, 2021, 3(1): 1-13.
|
| 24 |
ALHUMAIMESS Mosaed Saud, ALSOHAIMI Ibrahim Hotan, ALQADAMI Ayoub Abdullah, et al. Recyclable glutaraldehyde cross-linked polymeric tannin to sequester hexavalent uranium from aqueous solution[J]. Journal of Molecular Liquids, 2019, 281: 29-38.
|
| 25 |
MOHD Che Ku Nur Aima Che Ku, ISMAIL Mohd Halim Shah. Optimization of boron removal from aqueous solution via adsorption using composite beads of mangrove bark, alginate, and zeolite[J]. Journal of Chemical Engineering and Industrial Biotechnology, 2020, 3: 1-12.
|
| 26 |
ERDEM Pelin, BURSALI Elif Ant, YURDAKOC Muruvvet. Preparation and characterization of tannic acid resin: Study of boron adsorption[J]. Environmental Progress & Sustainable Energy, 2013, 32(4): 1036-1044.
|
| 27 |
G N Manjunatha REDDY, GERBEC Feffrey A, SHIMIZU Fumihiko, et al. Nanoscale surface compositions and structures influence boron adsorption properties of anion exchange resins[J]. Langmuir, 2019, 35(48): 15661-15673.
|
| 28 |
LI Meifeng, WANG Xiaoling, GONG Guidong, et al. Natural polyphenol-based nanoengineering of collagen-constructed hemoperfusion adsorbent for the excretion of heavy metals[J]. Journal of Hazardous Materials, 2022, 428: 128145.
|
| 29 |
BANGARI Raghubeer S, SINHA Niraj. Adsorption of tetracycline, ofloxacin and cephalexin antibiotics on boron nitride nanosheets from aqueous solution[J]. Journal of Molecular Liquids, 2019, 293: 111376.
|
| 30 |
LI Ping, LIU Chuang, ZHANG Li, et al. Enhanced boron adsorption onto synthesized MgO nanosheets by ultrasonic method[J]. Ultrasonics Sonochemistry, 2017, 34: 938-946.
|
| 31 |
SPRINGSTEEN Greg, WANG Binghe. A detailed examination of boronic acid-diol complexation[J]. Tetrahedron, 2002, 58(26): 5291-5300.
|
| 32 |
ALTUNINA L K, KUVSHINOV V A, STASIEVA L A, et al. The study of donor-acceptor equilibria in oil-displacing compositions based on acid coordinating solvents[J]. IOP Conference Series: Materials Science and Engineering, 2019, 597(1): 012017.
|
| 33 |
YAN Jun, SPRINGSTEEN Greg, DEETER Susan, et al. The relationship among pKa, pH, and binding constants in the interactions between boronic acids and diols—It is not as simple as it appears[J]. Tetrahedron, 2004, 60(49): 11205-11209.
|
| 34 |
Celine PONCET-LEGRAND, CABANE Bernard, Ana-Belen BAUTISTA-ORTÍN, et al. Tannin oxidation: intra-versus intermolecular reactions[J]. Biomacromolecules, 2010, 11(9): 2376-2386.
|
| 35 |
BARBEHENN Raymond V, JONES Christopher P, HAGERMAN Ann E, et al. Ellagitannins have greater oxidative activities than condensed tannins and galloyl glucose at high pH: Potential impact on caterpillars[J]. Journal of Chemical Ecology, 2006, 32(10): 2253-2267.
|
| 36 |
LUO Qinglong, ZENG Meiting, WANG Xueying, et al. Glycidol-functionalized macroporous polymer for boron removal from aqueous solution[J]. Reactive and Functional Polymers, 2020, 150: 104543.
|
| 37 |
WU Qiang, LIU Mingyan, WANG Xiong. A novel chitosan-based adsorbent for boron separation[J]. Separation and Purification Technology, 2019, 211: 162-169.
|
| 38 |
BERTAGNOLLI Caroline, GRISHIN Andrey, VINCENT Thierry, et al. Boron removal by a composite sorbent: Polyethylenimine/tannic acid derivative immobilized in alginate hydrogel beads[J]. Journal of Environmental Science and Health A, 2017, 52(4): 359-367.
|
| 39 |
BISHOP Maximilienne, SHAHID Naureen, YANG Jianzhong, et al. Determination of the mode and efficacy of the cross-linking of guar by borate using MAS 11B NMR of borate cross-linked guar in combination with solution 11B NMR of model systems[J]. Dalton Transactions, 2004(17): 2621-2634.
|
| 40 |
SANFELIU Cristina, Ramon MARTÍNEZ-MÁÑEZ, Felix SANCENÓN, et al. 11B-MAS NMR approach to the boron adsorption mechanism on a glucose-functionalised mesoporous silica matrix[J]. Microporous and Mesoporous Materials, 2018, 266: 232-241.
|
| 41 |
GRIMME Stefan, ANTONY Jens, EHRLICH Stephan, et al. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu[J]. The Journal of Chemical Physics, 2010, 132(15): 154104.
|
| 42 |
WEIGEND Florian, AHLRICHS Reinhart. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy[J]. Physical Chemistry Chemical Physics, 2005, 7(18): 3297-3305.
|
| 43 |
SONODA Akinari, MAKITA Yoji, Kenta OOI, et al. pH-dependence of the fractionation of boron isotopes with N-methyl-D-glucamine resin in aqueous solution systems[J]. The Chemical Society of Japan, 2000, 73(5): 1131-1133.
|
| 44 |
OI T, SHIMAZAKI H, ISHII R, et al. Boron isotope fractionation in liquid chromatography with boron-specific resins as column packing material[J]. Separation Science and Technology, 1997, 32(11): 1821-1834.
|
 ), 王晓玲1,2, 廖学品1,2,3(
), 王晓玲1,2, 廖学品1,2,3( ), 石碧1,2,3
), 石碧1,2,3
 ), WANG Xiaoling1,2, LIAO Xuepin1,2,3(
), WANG Xiaoling1,2, LIAO Xuepin1,2,3( ), SHI Bi1,2,3
), SHI Bi1,2,3