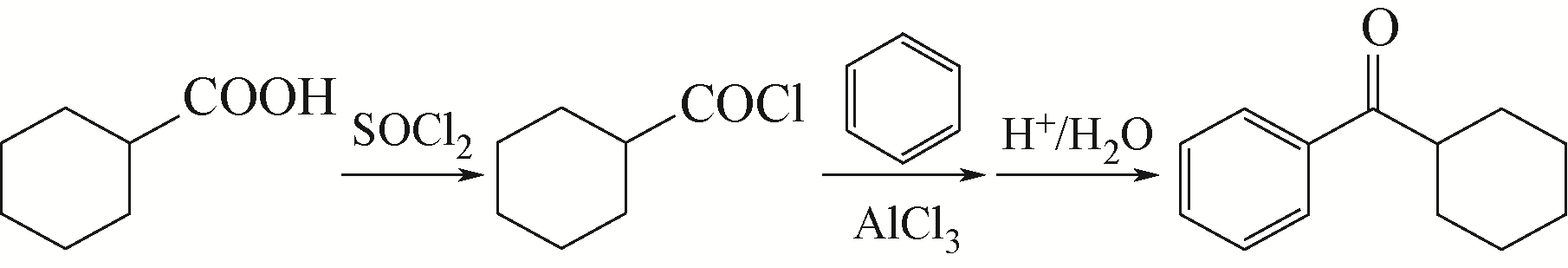| 1 |
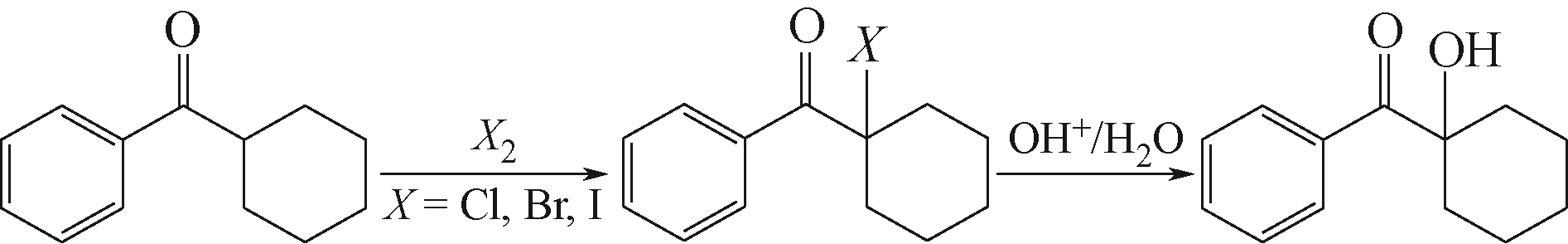
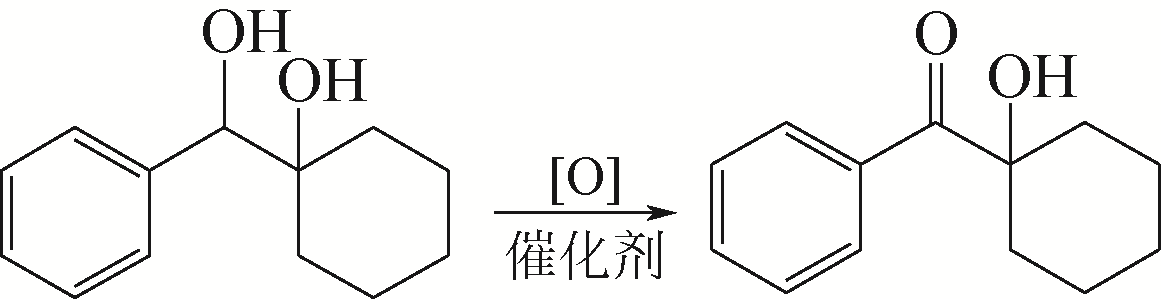
胡应喜, 陈赤阳, 刘霞, 等. α-羟基环己基苯基甲酮的合成[J]. 精细石油化工, 2005(1): 50-52.
|
|
HU Y X, CHEN C Y, LIU X, et al. Synthesis of α-hydroxycyclohexyl phenyl ketone[J]. Speciality Petrochemicals, 2005(1): 50-52.
|
| 2 |
夏晓勇, 陈捷. 紫外光固化涂料用光引发剂的应用研究进展[J]. 广州化工, 2014, 42(22): 26-29.
|
|
XIAO X Y, CHEN J. Research status and progress of photoinitiators used for UV curing paints[J]. Guangzhou Chemical Industry, 2014, 42(22): 26-29.
|
| 3 |
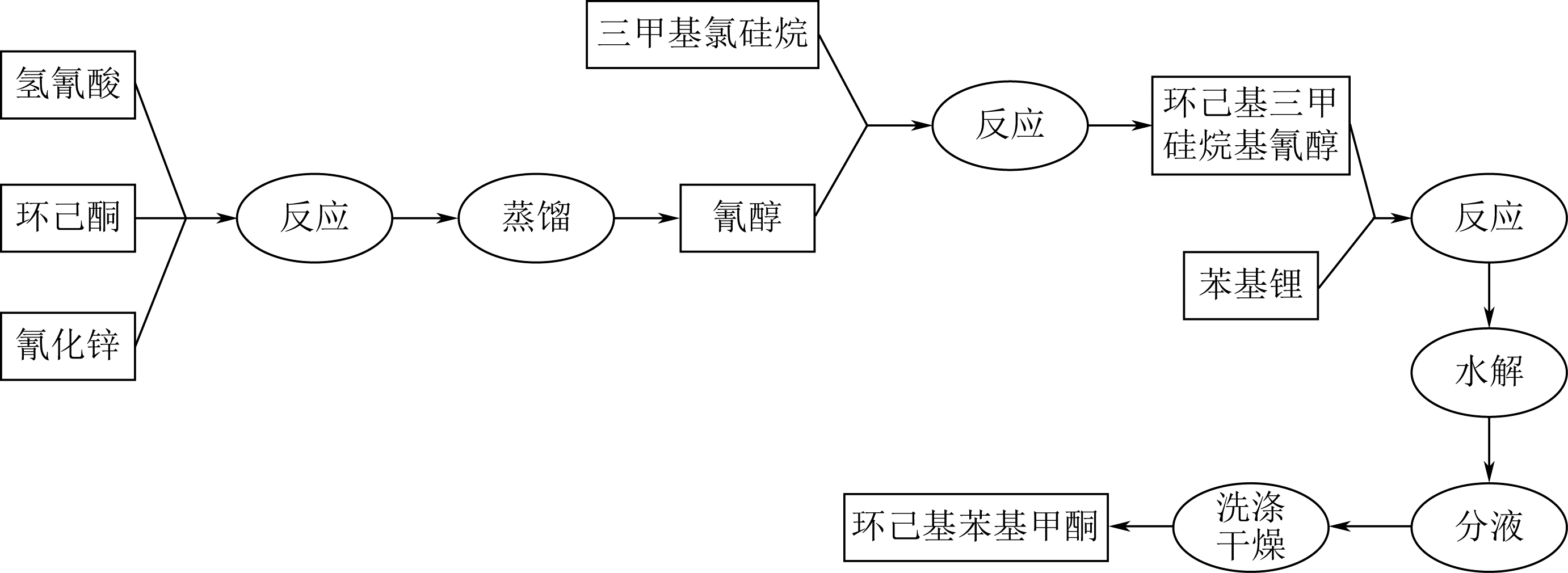
夏俊, 吴剑, 罗和安, 等. 1-羟基环己基苯基甲酮合成新工艺[J]. 化工时刊, 2004, 18(8): 49-50.
|
|
XIA J, WU J, LUO H A, et al. A new process for synthesizing 1-hydroxycyclohexyl phenyl ketone[J]. Chemical Industry Times, 2004, 18(8): 49-50.
|
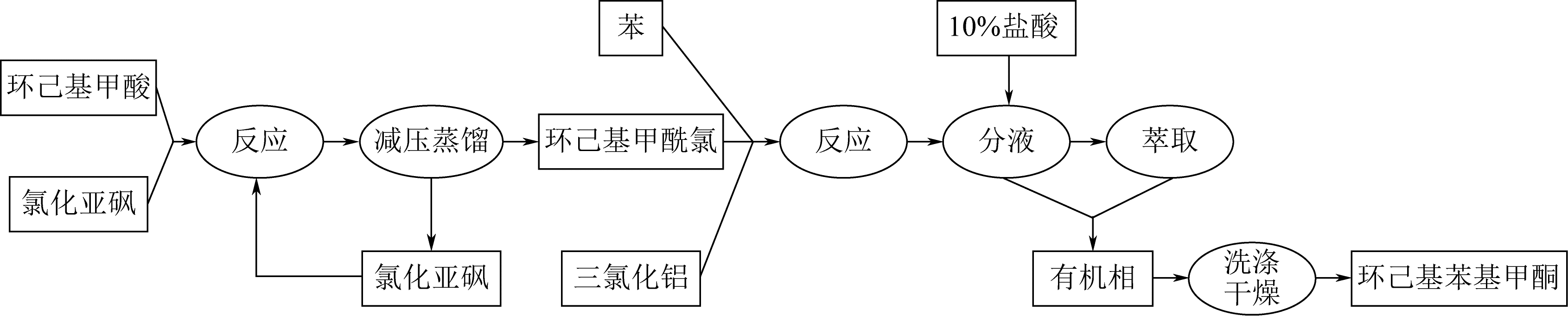
| 4 |
STEVENS C L, FARKAS E. The formation of 1-phenylcyclohexanecarboxylic acid from α-halocyclohexyl phenyl ketones[J]. Journal of the American Chemical Society, 1952, 74(21): 5352-5355.
|
| 5 |
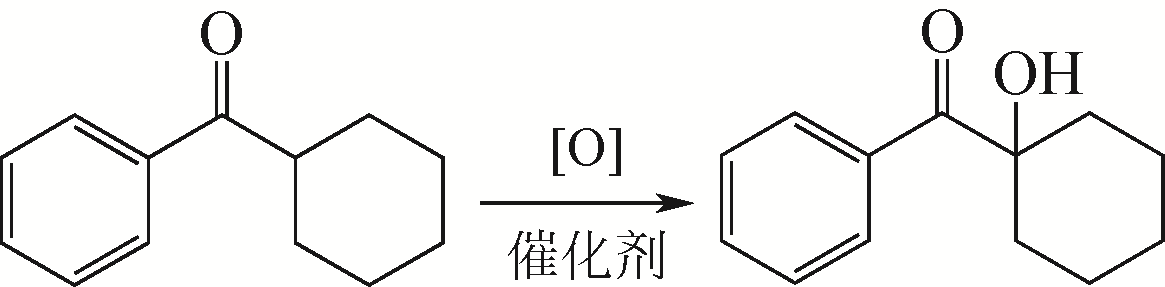
MENEGUZZO E, LI B G, NORCINI G. Procedure for the preparation of aromatic α-hydroxyketones: CN102015603[P]. 2011-04-13.
|
| 6 |
焦宁, 梁雨锋.一种α-羟基羰基化合物的合成方法: CN103613478[P]. 2015-05-06.
|
|
JIAO N, LIANG Y F. Preparation of α-hydroxycarbonyl compound: CN103613478[P]. 2015-05-06.
|
| 7 |
武瑞, 张齐, 罗想, 等. 一种α-羟基酮类光引发剂的制备方法: CN107739303[P]. 2018-02-27.
|
|
WU R, ZHANG Q, LUO X, et al. Method for preparing α-hydroxy ketone: CN107739303[P]. 2018-02-27.
|
| 8 |
REINHARD S, THOMAS L. Process for the preparation of alpha-functionalized ketones: WO197325[P]. 2018-11-01.
|
| 9 |
杨建明, 曲红梅, 邱历, 等. 光引发剂1-羟基环己基苯基甲酮的合成方法: CN108299172[P]. 2018-07-20.
|
|
YANG J M, QU H M, QIU L, et al. Method for synthesizing photoinitiator 1-hydroxycyclohexyl phenyl ketone: CN108299172[P]. 2018-07-20.
|
| 10 |
丁成荣, 陈林青, 王友昌, 等. 1-羟基环己基苯基甲酮合成新工艺研究[J]. 浙江工业大学学报, 2009, 27(3):263-266.
|
|
DING C R, CHEN L Q, WANG Y C, et al. Study on a new process for synthesis of 1-hydroxycyclohexyl phenyl methanone[J]. Journal of Zhejiang University of Technology, 2009, 27(3): 263-266.
|
| 11 |
HO B, CRIDER A M, STABLES J P. Synthesis and structure-activity relationships of potential anticonvulsants based on 2-piperidinecarboxylic acid and related pharmacophores[J]. European Journal of Medicinal Chemistry, 2001, 36(3): 265-286.
|
| 12 |
秦引林, 朱崇泉, 曹庆先. 氰基吡咯烷和氰基噻唑烷衍生物: CN101284810[P]. 2008-10-15.
|
|
QIN Y L, ZHU C Q, CAO Q X. Cyanopyrrolidine and cyanothiazolidine derivatives: CN101284810[P]. 2008-10-15.
|
| 13 |
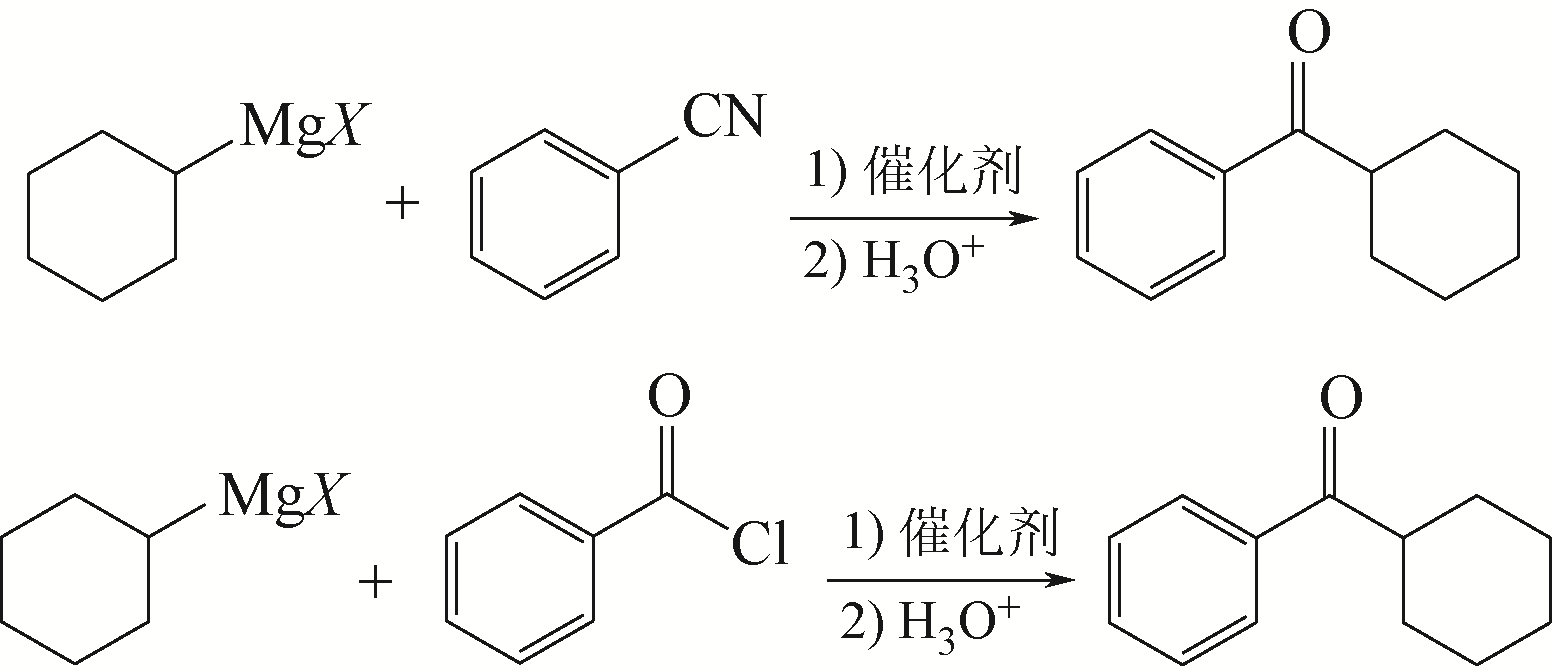
WEIBERTH F J, HALL S S. Copper(Ⅰ)-activated addition of Grignard reagents to nitriles. Synthesis of ketimines, ketones, and amines[J]. Journal of Organic Chemistry, 1987, 52(17): 3901-3904.
|
| 14 |
MALANGA C, ARONICA L A, LARDICCI L. Direct Ni mediated synthesis of ketones from acyl bromides and Grignard reagents[J]. Tetrahedron Letters, 1995, 36(50): 9185-9188.
|
| 15 |
FUKUMOTO Y, CHATANI N. e-EROS encyclopedia of reagents for organic synthesis[M]. New Jersey: John Wiley and Sons. Inc., 2005: 1-3.
|
| 16 |
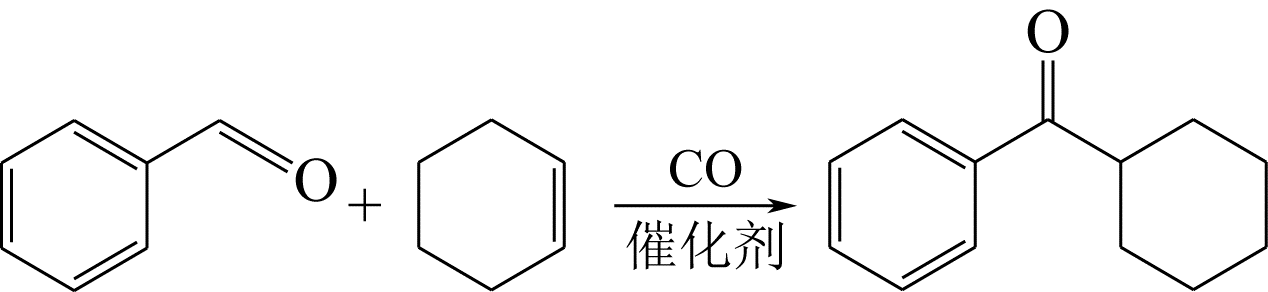
KONDO T, AKAZOME M, TSUJI Y, et al. Ruthenium complex catalyzed intermolecular hydroacylation and transhydroformylation of olefins with aldehydes[J]. Journal of Organic Chemistry, 1990, 55(4): 1286-1291.
|
| 17 |
YAÑEZ X, CLAVER C, CASTILLON S, et al. Montmorillonite K10 as a suitable co-catalyst for atom economy in chelation-assisted intermolecular hydroacylation[J]. Tetrahedron Letters, 2003, 44(8): 1631-1634.
|
| 18 |
罗邵伟, 吴剑, 罗和安. 一步法合成环己基苯基甲酮[J]. 湘潭大学自然科学学报, 2006, 28(3):95-97.
|
|
LUO S W, WU J, LUO H A. Synthesis of cyclohexyl phenyl ketone in one step[J]. Natural Science Journal of Xiangtan University, 2006, 28(3): 95-97.
|
| 19 |
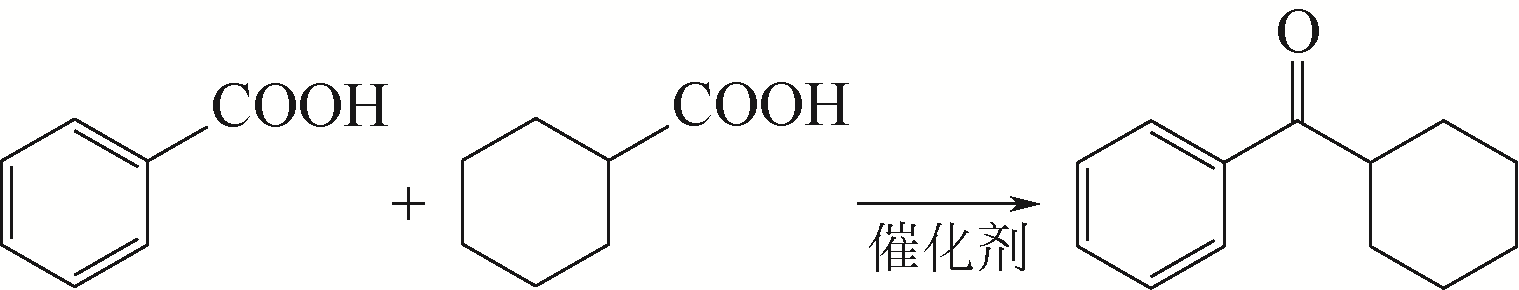
SCHOMMER C, EBEL K, DOCKNER T, et al. Preparation of ketones by dehydrative decarboxylation of carboxylic acids: EP0352674[P]. 1990-01-31.
|
| 20 |
张建锋, 武瑞, 张齐, 等. 一种羟基酮类光引发剂中间体芳基烷基酮的制备方法: CN107778155[P]. 2018-03-09.
|
|
ZHANG J F, WU R, ZHANG Q, et al. A method for preparing hydroxyketone photoinitiator intermediate arylalkyl ketone: CN107778155[P]. 2018-03-09.
|
| 21 |
薛圣耀, 陈志安, 林佑涵, 等. 制备芳基酮的方法: CN106431799[P]. 2017-02-22.
|
|
HSUEH S Y, CHEN Z A, LIN Y H, et al. Process for aryl ketone: CN106431799[P]. 2017-02-22.
|
| 22 |
石彩云. α-羟基环已基苯甲酮的合成方法: CN1281845[P]. 2001-1-31.
|
|
SHI C Y. Synthesis of α-hydroxycyclohexyl phenyl ketone: CN1281845[P]. 2001-1-31.
|
| 23 |
CHEN T, WANG Y. Environmentally-friendly new oxidation process for converting aryl-1,2-diol to ketone: WO2012100403[P]. 2012-08-02.
|
| 24 |
陈婷, 王瑜. 芳基取代邻二醇转化为羟基酮的可见光光促氧化工艺: CN103980101[P]. 2014-08-13.
|
|
CHEN T, WANG Y. Preparation process of hydroxy-ketone from aryl substituted vicinal diol via visible light photocatalytic oxidation reaction: CN103980101[P]. 2014-08-13.
|
| 25 |
HANSIORG G, HARTMUT S, SOUAD B. Process for converting alcohols to carbonyl compounds: US0203315[P]. 2005-09-15.
|
| 26 |
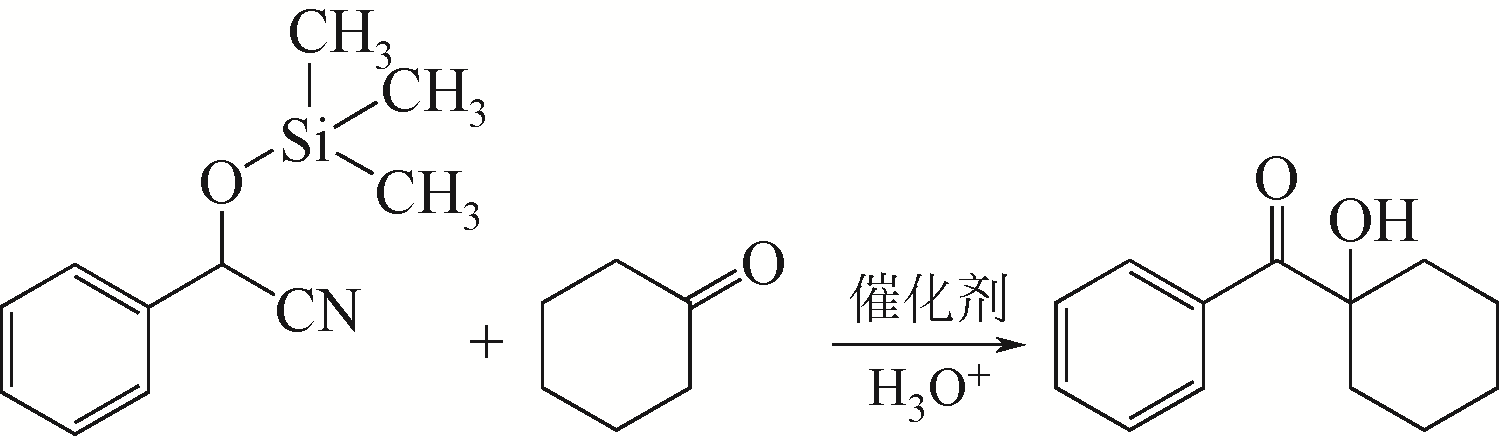
OHTA S, YAMASHITA M, ARITA K, et al. Conversion of ketone trimethylsilylcyanohydrins to several types of compounds[J]. Cheminform, 1995, 43(8): 1294-1301.
|
| 27 |
OLAH G A, WU A H. Trifluoromethanesulfonic(triflic) acid catalyzed transformations of alpha-hydroxy carbonyl compounds[J]. The Journal of Organic Chemistry, 1991, 56(7): 2531-2534.
|
| 28 |
KOENIGKRAMER R E, ZIMMER H. α-Heterosubstituted phosphonate carbanions Ⅸ: diethyl 1-phenyl-1-trimethylsiloxymethane phosphonate as an acyl anion equivalent; a novel method for the preparation of α-hydroxyketones[J]. Tetrahedron Letters, 1980, 21(11): 1017-1020.
|
| 29 |
HUENIG S, WEHNER G. Trimethylsilyl cyanide as an umpolung reagent. Ⅱ. Nucleophilic acylation of aldehydes and ketones with anionic 1,4-O,O-silyl group migration[J]. Chemischer Informationsdienst, 1979, 10(35): 2062-2067.
|
| 30 |
STEVEN M H, LARRY R K, JERALD K R. Process for preparing aryl ketone: US4524221[P]. 1985-06-18.
|
| 31 |
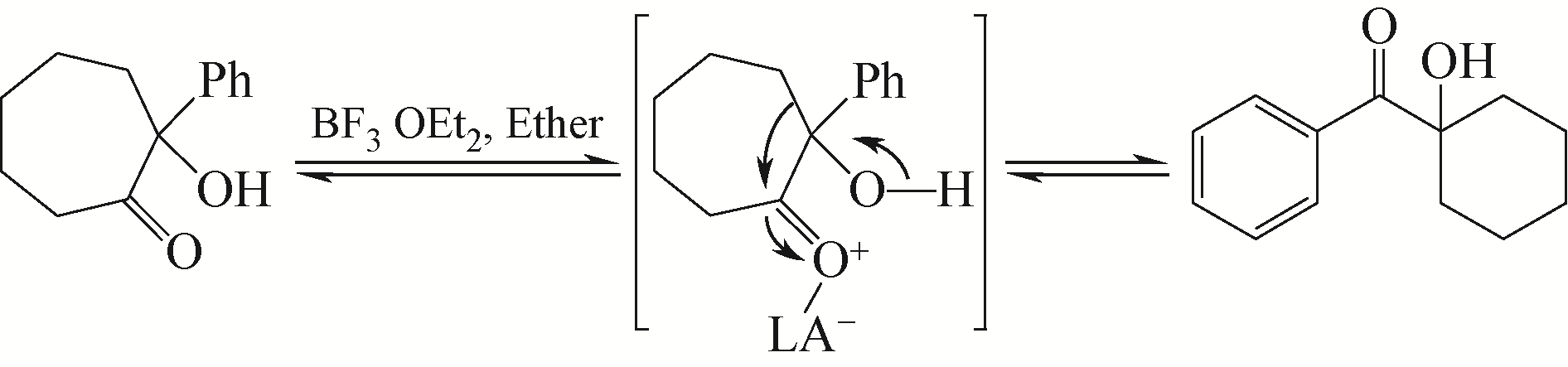
PAQUETTE L A, HOFFERBERTH J E. The α‐hydroxy ketone (α‐ketol) and related rearrangements[J]. Cheminform, 2005, 36(12): 525-528.
|
| 32 |
ELPHIMOFF F I, TCHOUBAR B. Expansion and contraction of carbon rings by isomerization of cyclic ketols[J]. Compt. Rend., 1953, 236: 1978-1980.
|
| 33 |
王辉明. 一种芳香族α-羟基酮化合物的合成方法: CN101225029[P]. 2008-07-23.
|
|
WANG H M. Process for preparation of aromatic α-hydroxy ketones: CN101225029[P]. 2008-07-23.
|
| 34 |
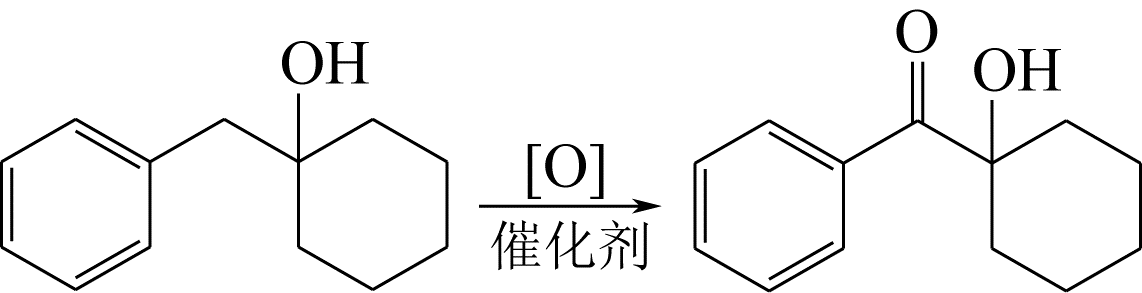
LYLE G G, COVEY R A, LYLE R E. Molecular rearrangements. Ⅳ. The pinacol rearrangement of 1-hydroxy-1-cyclohexylmethylphenylcarbinol[J]. Journal of the American Chemical Society, 1954, 76(10): 2713-2715.
|
| 35 |
CLERICI A, PORTA O. Reductive coupling of benzoyl cyanide and carbonyl compounds by aqueous titanium(Ⅲ) ions. A new convenient and selective access to the less stable mixed benzoins[J]. The Journal of Organic Chemistry, 1993, 58(10):2889-2893.
|
| 36 |
IRENE E F, MICHELE V. Preparation and spectral (UV and IR) properties of some isomeric pairs of α-ketols having or not having an aryl group conjugated with the carbonyl group[J]. Bulletin de la Societe Chimique de France, 1967, 3: 1047-1052.
|
| 37 |
HSUEH S Y, CHEN C A, LIN Y H, et al. Process for preparing aryl ketone: US0036981[P]. 2017-02-09.
|
| 38 |
朱骏飞, 周美华. 光引发剂的生产现状[J]. 精细与专用化学品, 2003, 19: 6-8.
|
|
ZHU J F, ZHOU M H. Production status of photoinitiators[J]. Fine and Specialty Chemicals, 2003, 19: 6-8.
|
| 39 |
周远航. 羟基酮类光引发剂合成工艺研究[D]. 南京: 南京理工大学, 2010.
|
|
ZHOU Y H. Study on a process for synthesis of α-hydroxy carbonyls photoinitiator[D]. Nanjing: Nanjing University of Science and Technology, 2010.
|
 ),Ze XU1,Jiancheng RUAN1,Xinzhi CHEN1,2(
),Ze XU1,Jiancheng RUAN1,Xinzhi CHEN1,2( )
)