Chemical Industry and Engineering Progress ›› 2021, Vol. 40 ›› Issue (9): 5214-5221.DOI: 10.16085/j.issn.1000-6613.2021-0553
Previous Articles Next Articles
Chiral NAD analogs as cofactors for biocatalysis
LI Qing1,2( ), LIU Wujun1, GUO Xiaojia1, WANG Qian1, ZHAO Zongbao1(
), LIU Wujun1, GUO Xiaojia1, WANG Qian1, ZHAO Zongbao1( )
)
- 1.Laboratory of Biotechnology, Dalian Institute of Chemical Physicals, CAS, Dalian 116023, Liaoning, China
2.University of Chinese Academy of Sciences, Beijing 100049, China
-
Received:2021-03-30Revised:2021-04-29Online:2021-09-13Published:2021-09-05 -
Contact:ZHAO Zongbao
手性NAD类似物合成及其辅酶应用
李青1,2( ), 刘武军1, 郭潇佳1, 王倩1, 赵宗保1(
), 刘武军1, 郭潇佳1, 王倩1, 赵宗保1( )
)
- 1.中国科学院大连化学物理研究所生物技术研究部,辽宁 大连 116023
2.中国科学院大学,北京 100049
-
通讯作者:赵宗保 -
作者简介:李青(1990—),女,博士研究生,研究方向为化学生物学。E-mail:liqing1910@dicp.ac.cn 。 -
基金资助:国家自然科学基金(21877112);国家重点研发计划(2019YFA0904900)
CLC Number:
Cite this article
LI Qing, LIU Wujun, GUO Xiaojia, WANG Qian, ZHAO Zongbao. Chiral NAD analogs as cofactors for biocatalysis[J]. Chemical Industry and Engineering Progress, 2021, 40(9): 5214-5221.
李青, 刘武军, 郭潇佳, 王倩, 赵宗保. 手性NAD类似物合成及其辅酶应用[J]. 化工进展, 2021, 40(9): 5214-5221.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: https://hgjz.cip.com.cn/EN/10.16085/j.issn.1000-6613.2021-0553
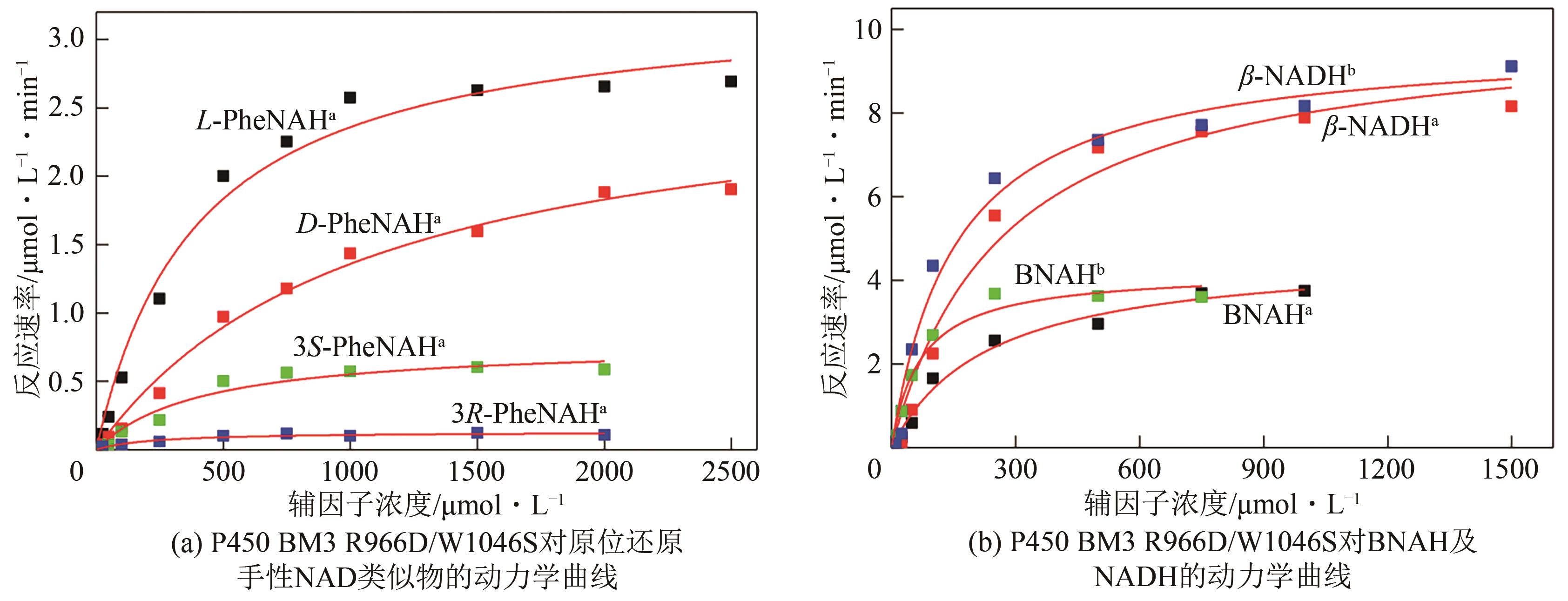
| 辅因子 | Km/mmol?L-1 | Kcat/s-1 | (Kcat?Km)/mmol-1?L?s-1 |
|---|---|---|---|
| L-PheNAH① | 0.40±0.06 | 0.07±0.00 | 0.17±0.02 |
| D-PheNDH① | 1.06±0.13 | 0.06±0.00 | 0.05±0.00 |
| 3S-PheNAH① | 0.41±0.11 | 0.02±0.00 | 0.04±0.01 |
| 3R-PheNAH① | 0.22±0.07 | 0.00±0.00 | 0.01±0.00 |
| BNAH① | 0.23±0.04 | 0.10±0.01 | 0.42±0.05 |
| β-NADH① | 0.27±0.06 | 0.21±0.01 | 0.78±0.12 |
| BNAH② | 0.07±0.01 | 0.09±0.00 | 1.25±0.11 |
| β-NADH② | 0.16±0.03 | 0.20±0.01 | 1.31±0.18 |
| 辅因子 | Km/mmol?L-1 | Kcat/s-1 | (Kcat?Km)/mmol-1?L?s-1 |
|---|---|---|---|
| L-PheNAH① | 0.40±0.06 | 0.07±0.00 | 0.17±0.02 |
| D-PheNDH① | 1.06±0.13 | 0.06±0.00 | 0.05±0.00 |
| 3S-PheNAH① | 0.41±0.11 | 0.02±0.00 | 0.04±0.01 |
| 3R-PheNAH① | 0.22±0.07 | 0.00±0.00 | 0.01±0.00 |
| BNAH① | 0.23±0.04 | 0.10±0.01 | 0.42±0.05 |
| β-NADH① | 0.27±0.06 | 0.21±0.01 | 0.78±0.12 |
| BNAH② | 0.07±0.01 | 0.09±0.00 | 1.25±0.11 |
| β-NADH② | 0.16±0.03 | 0.20±0.01 | 1.31±0.18 |
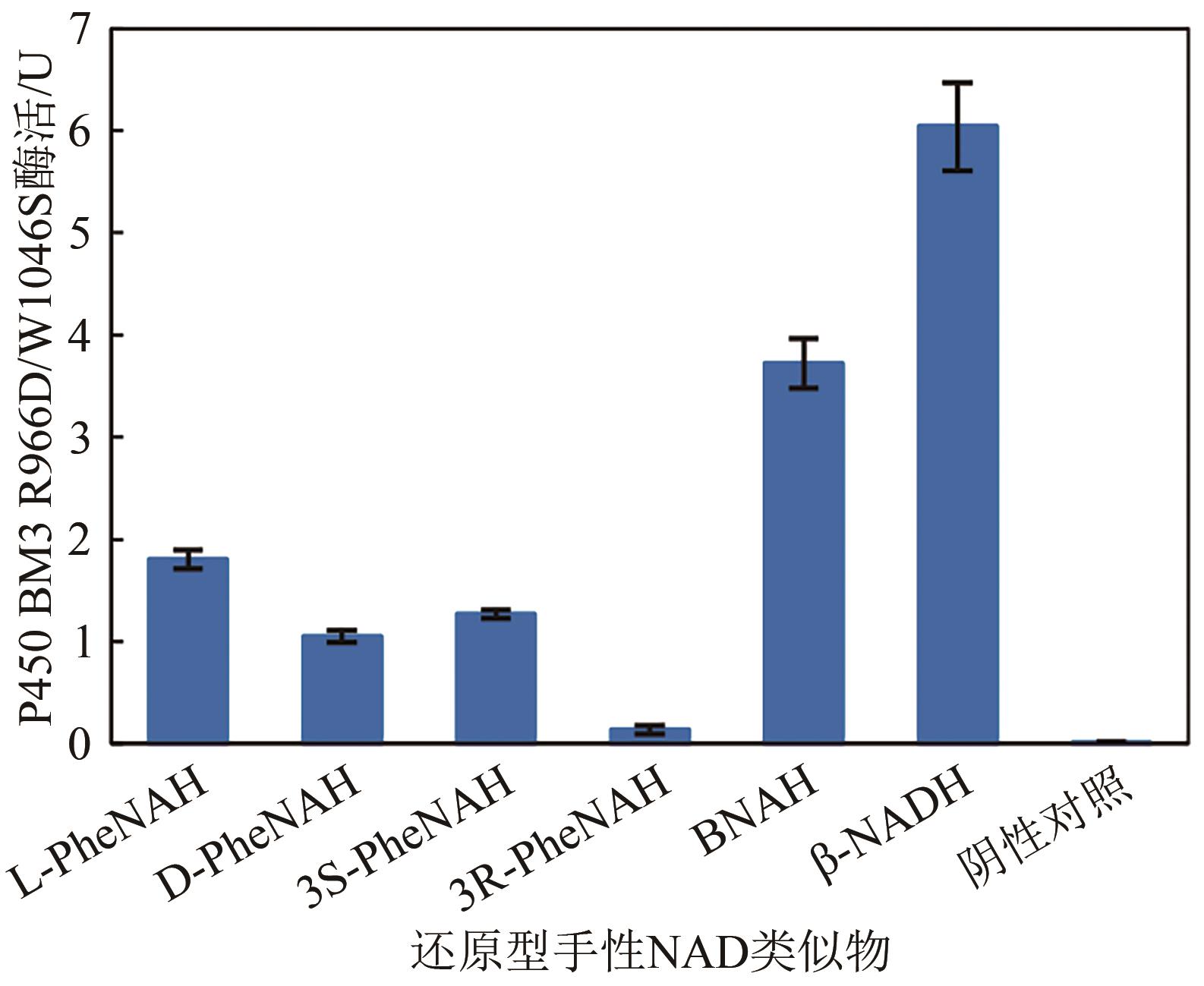
| 辅因子 | 酶活/U?mg-1 | 辅因子 | 酶活/U?mg-1 |
|---|---|---|---|
| L-PheNA | — | D-PheNA | — |
| 3S-PheNA | — | 3R-PheNA | — |
| BNA | 1.7×10-3 | β-NAD | 0.84 |
| 辅因子 | 酶活/U?mg-1 | 辅因子 | 酶活/U?mg-1 |
|---|---|---|---|
| L-PheNA | — | D-PheNA | — |
| 3S-PheNA | — | 3R-PheNA | — |
| BNA | 1.7×10-3 | β-NAD | 0.84 |
| 1 | XIAO W S, WANG R S, HANDY D E, et al. NAD(H) and NADP(H) redox couples and cellular energy metabolism[J]. Antioxidants & Redox Signaling, 2018, 28(3): 251-272. |
| 2 | MIYAKE Y, NAKAMURA Y, TAKAYAMA N, et al. Alpha reduced nicotinamide adenine dinucleotide-dependent reductase reactions of rat liver microsomes[J]. Journal of Biochemistry, 1975, 78(4): 773-783. |
| 3 | IZAGUIRRE G, PIETRUSZKO R, CHO S, et al. Human aldehyde dehydrogenase catalytic activity and structural interactions with coenzyme analogs[J]. Journal of Biomolecular Structure and Dynamics, 2001, 19(3): 429-447. |
| 4 | PARRY R J, HOYT J C. Purification and preliminary characterization of (E)-3-(2, 4-dioxo-6-methyl-5-pyrimidinyl)acrylic acid synthase, an enzyme involved in biosynthesis of the antitumor agent sparsomycin[J]. Journal of Bacteriology, 1997, 179(4): 1385-1392. |
| 5 | STEVENS L A, KATO J, KASAMATSU A, et al. The ARH and macrodomain families of α-ADP-ribose-acceptor hydrolases catalyze α-NAD+Hydrolysis[J]. ACS Chemical Biology, 2019, 14(12): 2576-2584. |
| 6 | SCHMIDT M T, SMITH B C, JACKSON M D, et al. Coenzyme specificity of sir2 protein deacetylases: implications for physiological regulation[J]. Journal of Biological Chemistry, 2004, 279(38): 40122-40129. |
| 7 | DE FLORA A, GUIDA L, FRANCO L, et al. Ectocellular in vitro and in vivo metabolism of cADP-ribose in cerebellum[J]. The Biochemical Journal, 1996, 320 (Pt 2): 665-671. |
| 8 | BEAUPRE B A, HOAG M R, CARMICHAEL B R, et al. Kinetics and equilibria of the reductive and oxidative half-reactions of human renalase with α-NADPH[J]. Biochemistry, 2013, 52(49): 8929-8937. |
| 9 | BEAUPRE B A, CARMICHAEL B R, HOAG M R, et al. Renalase is an α-NAD(P)H oxidase/anomerase[J]. Journal of the American Chemical Society, 2013, 135(37): 13980-13987. |
| 10 | COHEN M S, CHANG P. Insights into the biogenesis, function, and regulation of ADP-ribosylation[J]. Nature Chemical Biology, 2018, 14(3): 236-243. |
| 11 | PAUL C E, ARENDS I W C E, HOLLMANN F. Is simpler better? synthetic nicotinamide cofactor analogues for redox chemistry[J]. ACS Catalysis, 2014, 4(3): 788-797. |
| 12 | PAUL C E, HOLLMANN F. A survey of synthetic nicotinamide cofactors in enzymatic processes[J]. Applied Microbiology and Biotechnology, 2016, 100(11): 4773-4778. |
| 13 | ZACHOS I, NOWAK C, SIEBER V. Biomimetic cofactors and methods for their recycling[J]. Current Opinion in Chemical Biology, 2019, 49: 59-66. |
| 14 | HALLÉ F, FIN A, ROVIRA A R, et al. Emissive synthetic cofactors: enzymatic interconversions of tzA analogues of ATP, NAD+, NADH, NADP+, and NADPH[J]. Angewandte Chemie International Edition, 2018, 57(4): 1087-1090. |
| 15 | WANG L, JI D B, LIU Y X, et al. Synthetic cofactor-linked metabolic circuits for selective energy transfer[J]. ACS Catalysis, 2017, 7(3): 1977-1983. |
| 16 | ROVIRA A R, FIN A, TOR Y. Emissive synthetic cofactors: an isomorphic, isofunctional, and responsive NAD+ analogue[J]. Journal of the American Chemical Society, 2017, 139(44): 15556-15559. |
| 17 | DAI Z F, ZHANG X N, NASERTORABI F, et al. Facile chemoenzymatic synthesis of a novel stable mimic of NAD[J]. Chemical Science, 2018, 9(44): 8337-8342. |
| 18 | BLACK W B, ZHANG L Y, MAK W S, et al. Engineering a nicotinamide mononucleotide redox cofactor system for biocatalysis[J]. Nature Chemical Biology, 2020, 16(1): 87-94. |
| 19 | LIU W J, WU S G, HOU S H, et al. Synthesis of phosphodiester-type nicotinamide adenine dinucleotide analogs[J]. Tetrahedron, 2009, 65(40): 8378-8383. |
| 20 | 侯淑华, 刘武军, 赵宗保. 新型烟酰胺腺嘌呤二核苷酸(NAD)类似物的合成及其辅酶活性[J]. 有机化学, 2012, 32(2): 349-353. |
| HOU Shuhua, LIU Wujun, ZHAO Zongbao. Synthesis of novel nicotinamide adenine dinucleotide (NAD) analogs and their coenzyme activities[J]. Chinese Journal of Organic Chemistry, 2012, 32(2): 349-353. | |
| 21 | FRIEDLOS F, JARMAN M, DAVIES L C, et al. Identification of novel reduced pyridinium derivatives as synthetic co-factors for the enzyme DT diaphorase (NAD(P)H dehydrogenase (quinone), EC 1.6.99.2)[J]. Biochemical Pharmacology, 1992, 44(1): 25-31. |
| 22 | NOWAK C, PICK A, LOMMES P, et al. Enzymatic reduction of nicotinamide biomimetic cofactors using an engineered glucose dehydrogenase: providing a regeneration system for artificial cofactors[J]. ACS Catalysis, 2017, 7(8): 5202-5208. |
| 23 | LO H C, FISH R H. Biomimetic NAD+ models for tandem cofactor regeneration, horse liver alcohol dehydrogenase recognition of 1, 4-NADH derivatives, and chiral synthesis[J]. Angewandte Chemie, 2002, 114(3): 496-499. |
| 24 | RYAN J D, FISH R H, CLARK D S. Engineering cytochrome P450 enzymes for improved activity towards biomimetic 1, 4-NADH cofactors[J]. ChemBioChem, 2008, 9(16): 2579-2582. |
| 25 | LUTZ J, HOLLMANN F, HO T V, et al. Bioorganometallic chemistry: biocatalytic oxidation reactions with biomimetic NAD+/NADH co-factors and [Cp*Rh(bpy)H]+ for selective organic synthesis[J]. Journal of Organometallic Chemistry, 2004, 689(25): 4783-4790. |
| 26 | OKAMOTO Y, KÖHLER V, PAUL C E, et al. Efficient in situ regeneration of NADH mimics by an artificial metalloenzyme[J]. ACS Catalysis, 2016, 6(6): 3553-3557. |
| 27 | PAUL C E, GARGIULO S, OPPERMAN D J, et al. Mimicking nature: synthetic nicotinamide cofactors for C=C bioreduction using enoate reductases[J]. Organic Letters, 2013, 15(1): 180-183. |
| 28 | MAKAROV M V, MIGAUD M E. Syntheses and chemical properties of β-nicotinamide riboside and its analogues and derivatives[J]. Beilstein Journal of Organic Chemistry, 2019, 15: 401-430. |
| 29 | MADERN J M, KIM R Q, MISRA M, et al. Synthesis of stable NAD+ mimics as inhibitors for the legionella pneumophila phosphoribosyl ubiquitylating enzyme SdeC[J]. ChemBioChem, 2020, 21(20): 2903-2907. |
| 30 | DEPAIX A, KOWALSKA J. NAD analogs in aid of chemical biology and medicinal chemistry[J]. Molecules, 2019, 24(22): 4187. |
| 31 | JI D, WANG L, HOU S, et al. Creation of bioorthogonal redox systems depending on nicotinamide flucytosine dinucleotide[J]. Journal of the American Chemical Society, 2011, 133(51): 20857-20862. |
| 32 | CHENG W C, KURTH M J. The zincke reaction. A review[J]. Organic Preparations and Procedures International, 2002, 34(6): 585-608. |
| 33 | WALT D R, FINDEIS M A, RIOS-MERCADILLO V M, et al. An efficient chemical and enzymic synthesis of nicotinamide adenine dinucleotide (NAD+)[J]. Journal of the American Chemical Society, 1984, 106(1): 234-239. |
| 34 | IKBAL M, CERCEAU C, GOFFIC F, et al. Synthesis of the two enantiomers of the carbocyclic analog of nicotinamide ribose and analysis of their biological properties[J]. European Journal of Medical Chemistry, 1989, 24(4): 415-420. |
| 35 | HOCKOVÁ D, HOLÝ A. Synthesis of some “abbreviated” NAD+ analogues[J]. Collection of Czechoslovak Chemical Communications, 1997, 62(6): 948-956. |
| 36 | HOCKOVÁ D, VOTAVOVÁ H, HOLÝ A. Synthesis and chiroptical properties of some abbreviated NAD+ analogues[J]. Tetrahedron: Asymmetry, 1995, 6(9): 2375-2384. |
| 37 | SCHWANEBERG U, SCHMIDT-DANNERT C, SCHMITT J, et al. A continuous spectrophotometric assay for P450 BM-3, a fatty acid hydroxylating enzyme, and its mutant F87A[J]. Analytical Biochemistry, 1999, 269(2): 359-366. |
| 38 | CARELLI V, LIBERATORE F, SCIPIONE L, et al. Dithionite adducts of pyridinium salts: regioselectivity of formation and mechanisms of decomposition[J]. Tetrahedron, 2005, 61(43): 10331-10337. |
| 39 | ZHANG R Z, XU Y, XIAO R. Redesigning alcohol dehydrogenases/reductases for more efficient biosynthesis of enantiopure isomers[J]. Biotechnology Advances, 2015, 33(8): 1671-1684. |
| [1] | LI Huahua, LI Yihang, JIN Beichen, LI Longxin, CHENG Shao’an. Research progress of Anammox bio-electrochemical coupling wastewater treatment system [J]. Chemical Industry and Engineering Progress, 2023, 42(5): 2678-2690. |
| [2] | MENG Lingding, MAO Menglei, LIAO Qiyong, MENG Zihui, LIU Wenfang. Recent advance in stability of carbonic anhydrase and formate dehydrogenase [J]. Chemical Industry and Engineering Progress, 2022, 41(S1): 436-447. |
| [3] | GAO Bo, FENG Xudong, LI Chun. Visual and high-throughput method for detecting the activity of aspartate transcarbamylase [J]. Chemical Industry and Engineering Progress, 2022, 41(4): 2054-2059. |
| [4] | ZHANG Yan, WANG Wei, XIE Rui, JU Xiaojie, LIU Zhuang, CHU Liangyin. Controllable fabrication of polymeric microparticles loaded with enzyme@ZIF-8 [J]. Chemical Industry and Engineering Progress, 2022, 41(4): 2022-2028. |
| [5] | TANG Wenxiu, WANG Xueming, GUO Liang, JI Lihao, GAO Cong, CHEN Xiulai, LIU Liming. Metabolic engineering of Escherichia coli to produce succinic acid [J]. Chemical Industry and Engineering Progress, 2022, 41(2): 938-950. |
| [6] | LU Zeping, PEI Xinhua, XUE Yu, ZHANG Xiaoguang, HU Yi. Chemical modification of porcine pancreatic lipase with betaine ionic liquid to improve its enzymatic properties [J]. Chemical Industry and Engineering Progress, 2022, 41(11): 6045-6052. |
| [7] | LI Qingyuan, WANG Chao, XU Shipei, ZHANG Xueqin, QIU Mingjian, LIU Mengyao, CONG Mengxiao. Research progress on reaction process and catalysts for PBS precursor of 1,4-butanediol synthesis [J]. Chemical Industry and Engineering Progress, 2022, 41(11): 5771-5782. |
| [8] | JU Shuyun, WU Jianping, YANG Lirong. Advances in the molecular modification and application of D-amino acid oxidase [J]. Chemical Industry and Engineering Progress, 2021, 40(3): 1215-1225. |
| [9] | ZHANG Xiaojian, LIU Qian, LIU Zhiqiang, ZHENG Yuguo. Stereoselective carbonyl reductases and their application in chiral alcohols synthesis [J]. Chemical Industry and Engineering Progress, 2021, 40(3): 1142-1160. |
| [10] | Zhufan LIN, Shao’an CHENG, Zhengzhong MAO, Ruonan GU, Jiawei YANG. Recent advances in the construction and influencing factors of bio-electrochemical nitrogen removal systems [J]. Chemical Industry and Engineering Progress, 2020, 39(9): 3766-3776. |
| [11] | Cheng ZHU,Guochao XU,Wei DAI,Jieyu ZHOU,Ye NI. Effect of position 127 on the activity and enantioselectivity of alcohol dehydrogenase KpADH [J]. Chemical Industry and Engineering Progress, 2019, 38(12): 5504-5511. |
| [12] | Tian JIANG, Xudong FENG, Yan LI, Chu LI. The biocatalysis and enzyme modification of substrate specificity [J]. Chemical Industry and Engineering Progress, 2019, 38(01): 606-614. |
| [13] | YU Bo, LIU Chao, LIU Jindong, DING Wanyu, CHAI Weiping. Preparation of mesoporous zirconium phosphate and its catalytic performace in the preparation of cellulose from glucose [J]. Chemical Industry and Engineering Progress, 2018, 37(06): 2236-2241. |
| [14] | YAN Xingchen, ZHAO Qianru, WANG Kaifeng, GUO Yuxin, JIANG Ling, HUANG He. Auto-induced expression of trehalose synthetase and novel process for catalytic production of trehalose [J]. Chemical Industry and Engineering Progress, 2018, 37(05): 1949-1955. |
| [15] | WANG Rui, XU Yaohui, WANG Kewei, WU Minchen. Expression of PvEH3,a Phaseolus vulgaris epoxide hydrolase,and synthesis of chiral vicinal diols [J]. Chemical Industry and Engineering Progress, 2018, 37(05): 1933-1939. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||