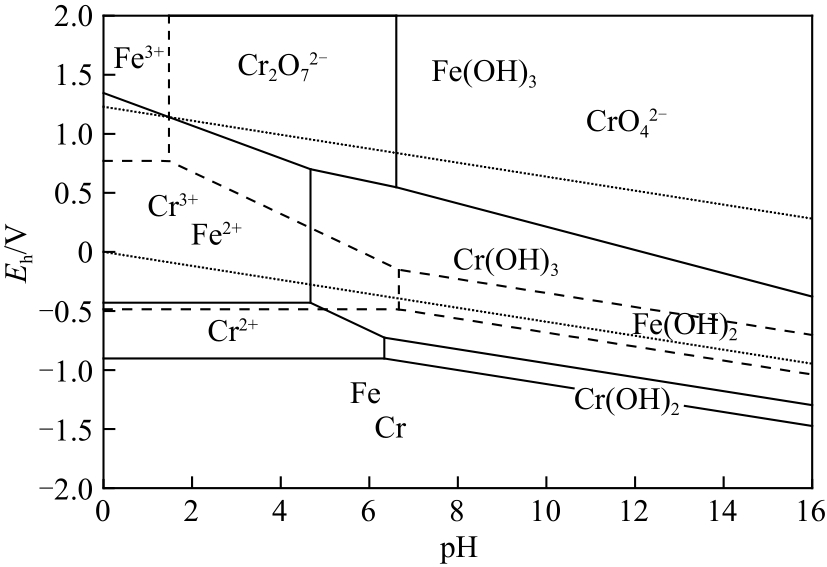
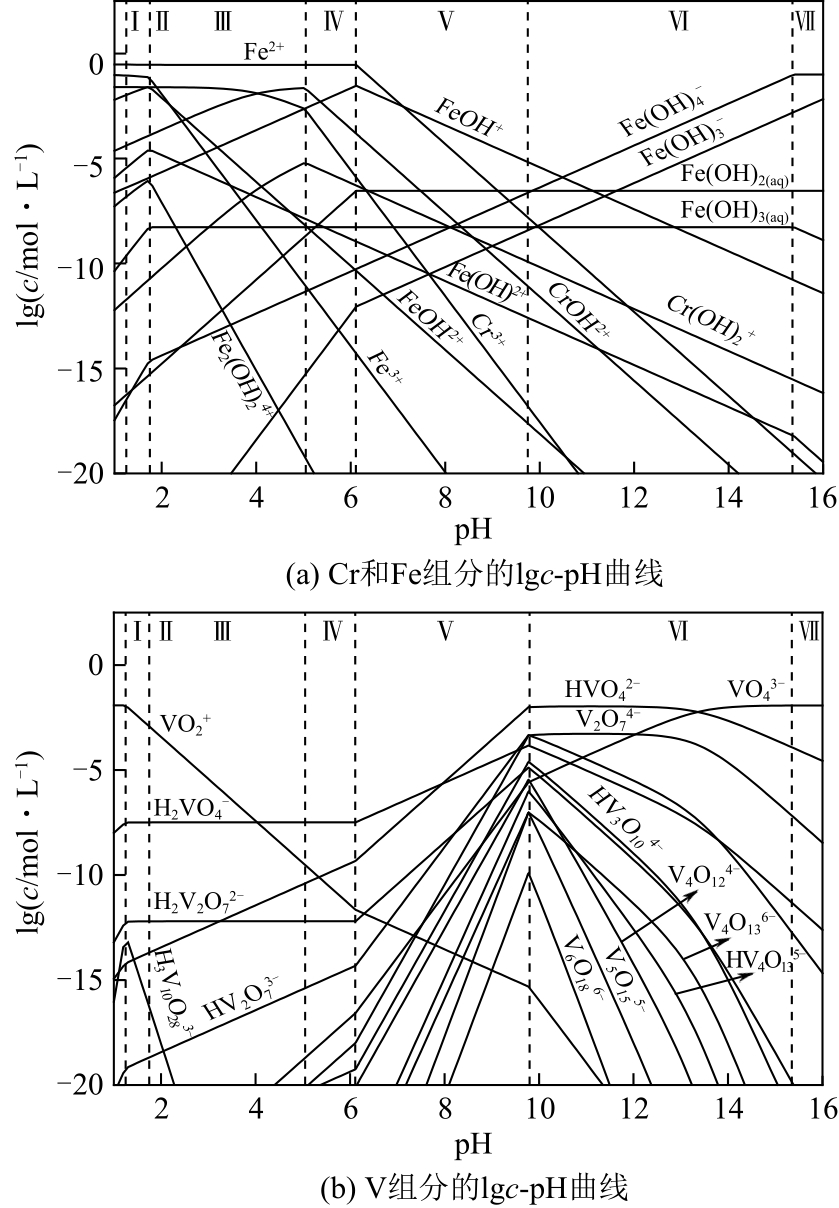
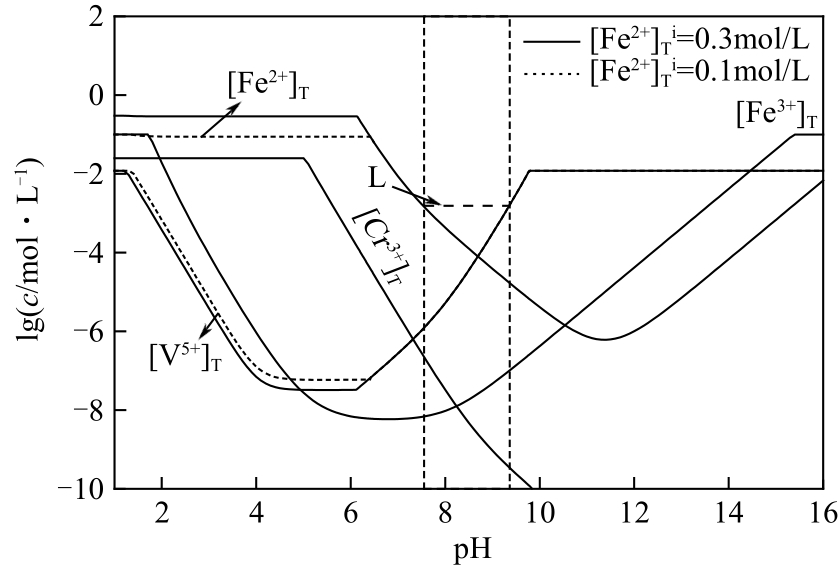
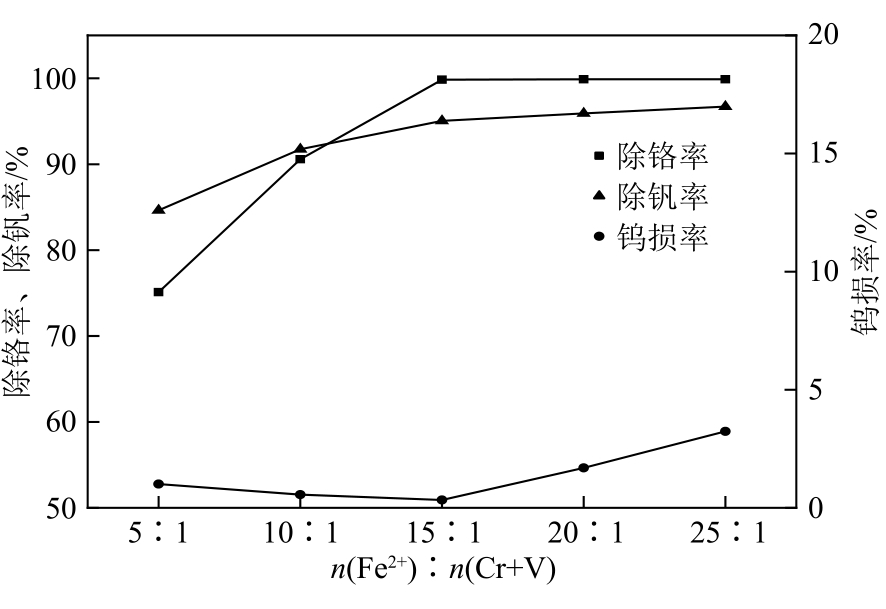
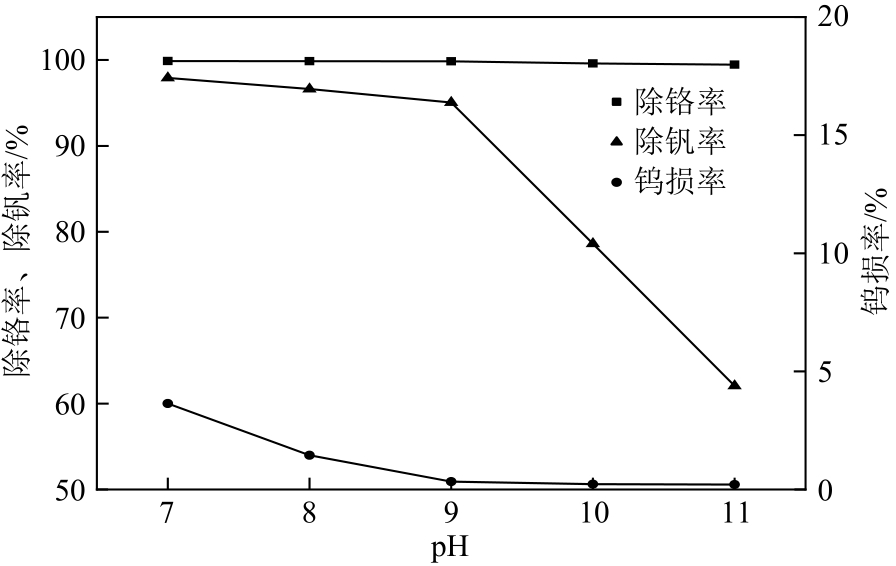
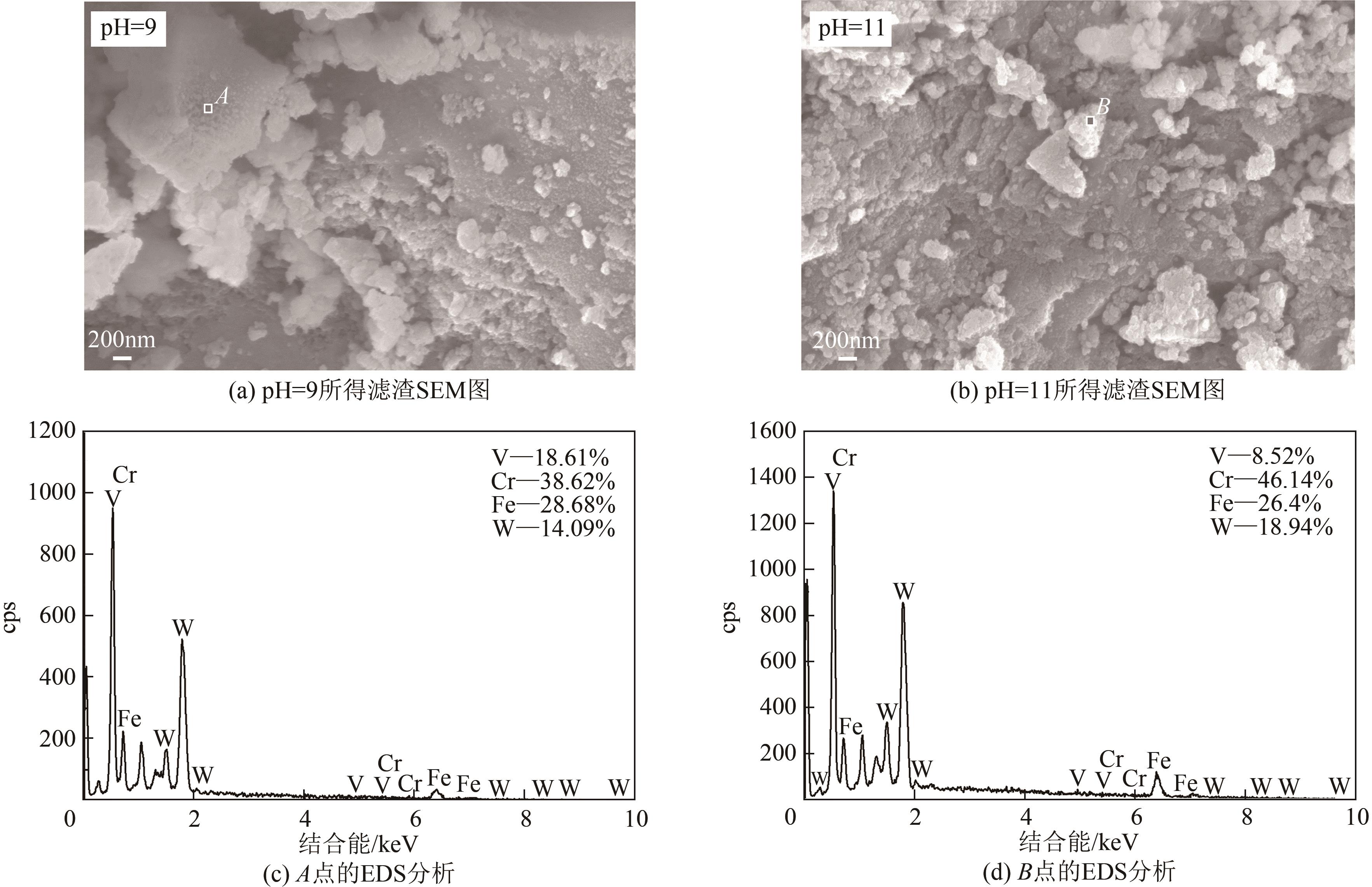
| 21 |
LI Xin, LI Ming, LIANG Bin. Removal of vanadium from acidic chromium solution by ferric sulfate[J]. Iron Steel Vanadium Titanium, 2016, 37(4): 20-24.
|
| 22 |
杨得军, 王少娜, 陈晓芳, 等. 铬盐无钙焙烧工艺铬酸钠中性液铁盐除钒[J]. 中国有色金属学报, 2014, 24(1): 279-285.
|
|
YANG Dejun, WANG Shaona, CHEN Xiaofang, et al. Removing vanadium from sodium chromate neutral liquid by non-calcium roasting technology with chromium salt[J]. The Chinese Journal of Nonferrous Metals, 2014, 24(1): 279-285.
|
| 23 |
刘芳. 还原沉淀法对含铬重金属废水的处理研究[J]. 环境污染与防治, 2014, 36(4): 54-59.
|
|
LIU Fang. Treatment of chromium containing heavy metal wastewater by reduction and sedimentation process[J]. Environmental Pollution & Control, 2014, 36(4): 54-59.
|
| 1 |
MISHRA D, SINHA S, SAHU K K, et al. Recycling of secondary tungsten resources[J]. Transactions of the Indian Institute of Metals, 2017, 70(2): 479-485.
|
| 2 |
周新华, 王力民, 彭英健. 我国硬质合金再生产业现状与发展[J]. 硬质合金, 2016, 33(5): 356-364.
|
|
ZHOU Xinhua, WANG Limin, PENG Yingjian. Status and development of cemented carbide recycling industry in China[J]. Cemented Carbide, 2016, 33(5): 356-364.
|
| 3 |
李洪桂. 稀有金属冶金学[M]. 北京: 冶金工业出版社, 1990: 22.
|
|
LI Honggui. Rare metals metallurgy[M]. Beijing: Metallurgical Industry Press, 1990: 22.
|
| 4 |
谢盛德, 李春海, 雷吟春. 废钨回收中钨酸钠结晶料的除铬处理研究与实践[J]. 稀有金属与硬质合金, 2012, 40(3): 8-12.
|
|
XIE Shengde, LI Chunhai, LEI Yinchun. Research and practice of chromium removal from sodium tungstate crystal material during waste tungsten recovery[J]. Rare Metals and Cemented Carbides, 2012, 40(3): 8-12.
|
| 5 |
雷吟春, 欧阳海波, 曹育龙, 等. 镁盐共沉淀法深度除铬及WO3的回收研究[J]. 稀有金属与硬质合金, 2019, 47(6): 1-4.
|
|
LEI Yinchun, OUYANG Haibo, CAO Yulong, et al. Study on deep chromium removal by magnesium salt coprecipitation and recovery of WO3 [J]. Rare Metals and Cemented Carbides, 2019, 47(6): 1-4.
|
| 6 |
张乐怡, 吴红梅, 姜晓庆, 等. 活性氧化铝的改性及除铬(Ⅵ)性能研究[J]. 天津化工, 2020, 34(1): 11-14.
|
|
ZHANG Leyi, WU Hongmei, JIANG Xiaoqing, et al. Modification of active alumina with sodium hydroxide for adsorption of chromium (Ⅵ) ions[J]. Tianjin Chemical Industry, 2020, 34(1): 11-14.
|
| 7 |
肖超, 曾理, 李义兵, 等. 磷酸盐沉淀法除铬热力学研究[J]. 有色金属科学与工程, 2017, 8(5): 103-108.
|
|
XIAO Chao, ZENG Li, LI Yibing, et al. Thermodynamic study on chromium removal by phosphate precipitation[J]. Nonferrous Metal Science and Engineering, 2017, 8(5): 103-108.
|
| 8 |
宋阜, 朱宾权. 离子交换法分离富集钨酸钠溶液中的钒[J]. 稀有金属与硬质合金, 2006, 34(3): 5-7, 11.
|
|
SONG Fu, ZU Binquan. Extraction of vanadium from sodium tungstate by ion exchange[J]. Rare Metals and Cemented Carbides, 2006, 34(3): 5-7, 11.
|
| 9 |
LUO Lin, KEJUN Liu, SHIBAYAMA Atsushi, et al. Recovery of tungsten and vanadium from tungsten alloy scrap[J]. Hydrometallurgy, 2004, 72(1): 1-8.
|
| 10 |
陈星宇, 肖露萍, 赵中伟. 钨钼冶炼过程中除钒[J]. 中国有色金属学报, 2014, 24(7): 1883-1887.
|
|
CHEN Xingyu, XIAO Luping, ZHAO Zhongwei. Vanadium removal in tungsten or molybdenum smelting process[J]. The Chinese Journal of Nonferrous Metals, 2014, 24(7): 1883-1887.
|
| 11 |
张家靓, 赵中伟. W(Ⅵ)-V(Ⅴ)-H2O体系钨钒分离的热力学分析[J]. 中国有色金属学报, 2014, 24(6): 1656-1662.
|
|
ZHANG Jialiang, ZHAO Zhongwei. Thermodynamic analysis for separation of tungsten and vanadium in W(Ⅵ)-V(Ⅴ)-H2O system[J]. The Chinese Journal of Nonferrous Metals, 2014, 24(6): 1656-1662.
|
| 12 |
SADIQ Muhammad. Thermodynamic solubility relationships of inorganic vanadium in the marine environment[J]. Marine Chemistry, 1988, 23(12): 87-96.
|
| 13 |
张文娟, 马保中, 王成彦. 基于磷酸盐沉淀分离电镀污泥中铬铁的热力学研究[J]. 稀有金属, 2018, 42(10): 1084-1092.
|
|
ZHANG Wenjuan, MA Baozhong, WANG Chengyan. Thermodynamic study on selective separation of chromium and iron from electroplating sludge based on phosphate precipitation[J]. Chinese Journal of Rare Metals, 2018, 42(10): 1084-1092.
|
| 14 |
VAGHETTI J C P, LIMA E C, ROYER B, et al. Application of Brazilian-pine fruit coat as a biosorbent to removal of Cr(Ⅵ) from aqueous solution—Kinetics and equilibrium study[J]. Biochemical Engineering Journal, 2008, 42(1): 67-76.
|
| 15 |
赵中伟. 钨冶炼的理论与应用[M]. 北京: 清华大学出版社, 2013: 75.
|
|
ZHAO Zhongwei. Tungsten metallurgy: Fundamentals and applications[M]. Beijing: Tsinghua University Press, 2013: 75.
|
| 16 |
CHANG Liyang. Alternative chromium reduction and heavy metal precipitation methods for industrial wastewater[J]. Environmental Progress, 2003, 22(3):174-182.
|
| 17 |
SPEIGHT J G. Lange's handbook of chemistry[M]. 16th ed. New York: McGraw-Hill Professional, 2005: 1237.
|
| 18 |
杨显万. 高温水溶液热力学数据计算手册[M]. 北京: 冶金工业出版社, 1983: 645.
|
|
YANG Xianwan. Handbook of thermodynamic data of high temperature aqueous solution[M]. Beijing: Metallurgical Industry Press, 1983: 645.
|
| 19 |
PEACOCK C L, SHERMAN D M. Vanadium(Ⅴ) adsorption onto goethite (α-FeOOH) at pH 1.5 to 12: A surface complexation model based on ab initio molecular geometries and EXAFS spectroscopy[J]. Geochimica et Cosmochimica Acta, 2004, 68(8): 1723-1733.
|
| 20 |
COLTON J V, MICHAEL P S, MOJTABA A, et al. Adsorption of (poly)vanadate onto ferrihydrite and hematite: An in situ ATR-FTIR study[J]. ACS Earth and Space Chemistry, 2020, 4(4): 641-649.
|
| 21 |
李信, 李明, 梁斌. 利用硫酸铁除去沉钒后酸性铬溶液中钒的试验研究[J]. 钢铁钒钛, 2016, 37(4): 20-24.
|
 ), 潘界昌1, 徐国钻2, 李文君1, 梁勇1(
), 潘界昌1, 徐国钻2, 李文君1, 梁勇1( )
)
 ), PAN Jiechang1, XU Guozuan2, LI Wenjun1, LIANG Yong1(
), PAN Jiechang1, XU Guozuan2, LI Wenjun1, LIANG Yong1( )
)